Reprogramming Blood Cells: How to Decrease Variability in Your Workflow
A Guest Blog by Wing Chang, PhD, Scientist, and Andrea Lai, PhD, Scientific Marketing Specialist, STEMCELL Technologies.
iPS Cells
Human induced pluripotent stem cells (iPS cells) have the potential to impact innumerable research and clinical applications such as disease modeling, drug discovery, toxicity screening or Cell Therapy. iPS cells are generated by reprogramming somatic cells to a pluripotent state, through the transient overexpression of key reprogramming factors.
The first human cell type to be converted to iPS cells were dermal fibroblasts, and these are still one of the most common sources used for reprogramming experiments.(1,2) Since then, other primary cell sources such as keratinocytes, mesenchymal stem cells, T-cells, B-cells, hematopoietic progenitors, and urine epithelial cells have also been reprogrammed to iPS cells.(1–8) The choice of starting cell type for iPS cell generation is influenced by factors such as availability of donor tissue from normal and diseased patients, invasiveness of sample collection procedures, genomic integrity, epigenetic memory, and reprogramming efficiency.
Peripheral Blood Cells for Reprogramming
Peripheral blood (PB) is a popular tissue source for generating iPS cells.(9) Blood collection is a well-established and minimally invasive procedure, and collected cells are naturally replaced as the tissue is self-renewing. Banked blood samples are also available for a wide variety of disease, age, gender and geographical subtypes. Since the cells are continually replenished from stem cells in the bone marrow, it is expected that they will contain fewer environment-associated point mutations than skin, which is exposed to long-term ultraviolet radiation.
However, PB contains a heterogeneous mixture of cell types, the most prevalent of which are enucleated (e.g. mature red blood cells (RBCs) and platelets) and therefore not suitable for reprogramming (to learn more about these cells and their frequencies, view our popular wallchart).
The first step in preparing whole PB samples for reprogramming is to separate the PB mononuclear cell (PBMC) fraction from the RBCs and platelets. This is generally done by density gradient centrifugation. The separated PBMC fraction contains T and B lymphocytes, macrophages, monocytes, erythroid progenitors and rare circulating stem cells. Many of these cell types have been successfully reprogrammed with varying efficiencies.(9) While T- and B-cells are the most abundant cell type in the PBMC fraction and have been successfully reprogrammed, they contain V(D)J genomic rearrangements of the T-cell receptor or immunoglobulin loci, respectively. The ability to generate iPS cells from specific T-cells has been utilized for proof of principle experiments in T-Cell Therapy,(10) but little is known about how these rearrangements may affect the function of other downstream cell lineages.
Less abundant cell types including CD34+ hematopoietic stem and progenitor cells, and erythroid progenitor cells are attractive for reprogramming, due to their lack of genomic rearrangements and demonstrated reprogramming ability.(7,11) However, owing to their low frequency in whole blood, these cells need to be isolated from PB and/or expanded in vitro to obtain sufficient cell numbers for reprogramming.
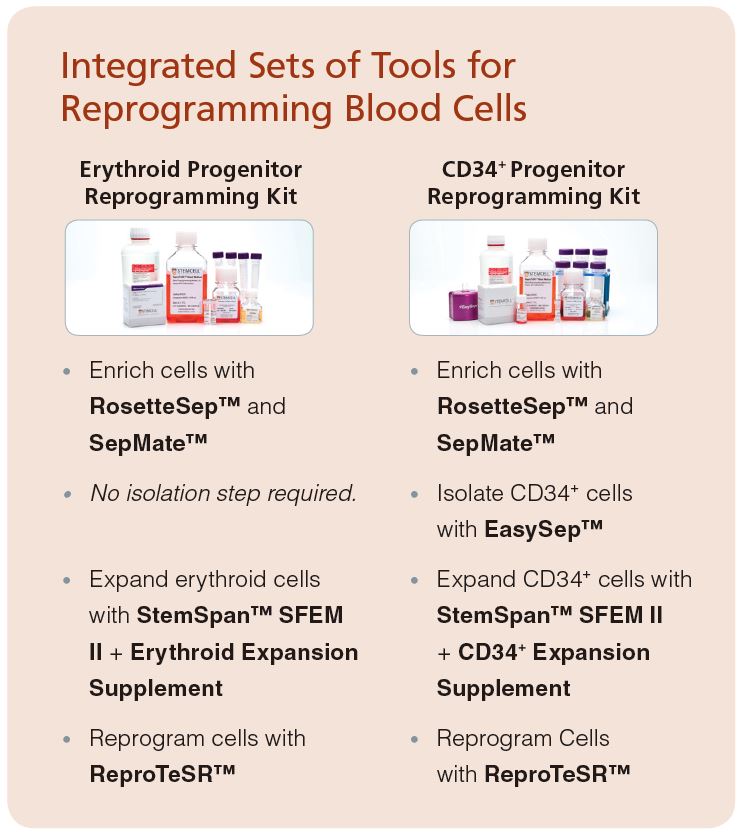
An Integrated Workflow
At STEMCELL Technologies, we have developed integrated sets of tools to standardize and facilitate reprogramming of erythroid or CD34+ progenitor cells to human iPS cells.
Rapid Cell Enrichment
As mentioned previously, successful generation of iPS cells from PB requires the removal of unwanted cells such as mature RBCs and platelets, and retention of the desired progenitor cell population. PBMC isolation is traditionally a lengthy and technically difficult procedure. Density gradient centrifugation can take over 45 minutes (with no break) and the challenging techniques of blood sample layering and PBMC interphase harvesting can lead to significant variability between users.
To better handle the challenges of harvesting the desired cells from blood the Erythroid Progenitor and CD34+ Progenitor Reprogramming Kits both include SepMate™ tubes. These tubes contain a unique insert that allows you to quickly pipet or pour blood over Lymphoprep™ density gradient medium without careful layering, as the insert prevents the layers from mixing. Centrifugation is cut down to just 10 minutes (with the brake on) and the enriched progenitor cells are simply poured into a fresh tube.
As previously stated, mature cell types such as T- and B-cells may not be suitable sources for reprogramming, due to the presence of V(D)J rearrangements. These cell types can be quickly depleted using RosetteSep™ (Figure 1). RosetteSep™ binds unwanted cells to RBCs, forming immunorosettes, which pellet during density gradient centrifugation. Use the RosetteSep™ Human Progenitor Cell Basic Pre-Enrichment Cocktail to remove T- and B-cells prior to erythroid cell expansion.
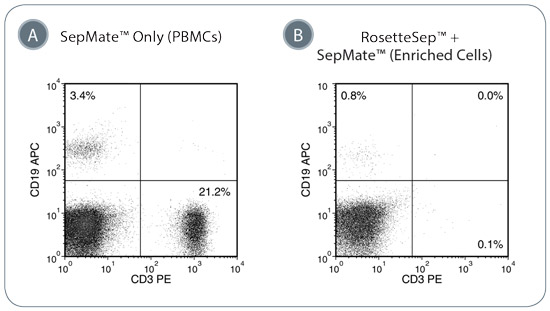
Figure 1. Depletion of T-Cells and B-Cells from Whole Blood with RosetteSep™ Human Progenitor Cell Basic Pre-Enrichment Cocktail and SepMate™ Tubes
(A) PBMCs were enriched from whole blood using SepMate™ only. In this example, T-cells (CD3+) represent approximately 21.2% of the PBMC fraction while B-cells (CD19+) are present at 3.4%. (B) RosetteSep™ and SepMate™ were used to deplete lineage-committed cells from blood. The RosetteSep™ Human Progenitor Cell Basic Pre-Enrichment cocktail efficiently depletes the T- and B-cell population to < 1% of the enriched cell fraction.
Immunomagnetic CD34+ Cell Isolation
The EasySep™ Complete Kit for Human Whole Blood CD34+ Cells is a simple method for isolating highly purified CD34+ stem and progenitor cells from blood (Figure 2). This kit contains both RosetteSep™ and EasySep™ components. First, the RosetteSep™ Human Hematopoietic Progenitor Enrichment Cocktail is used to deplete lineage-positive cells during density gradient centrifugation. Next, the CD34+ cells are labeled with the EasySep™ Human CD34 Positive Selection cocktail and magnetic particles, and separated without columns using an EasySep™ magnet. Unwanted cells are simply poured off, while CD34+ cells remain in the tube.
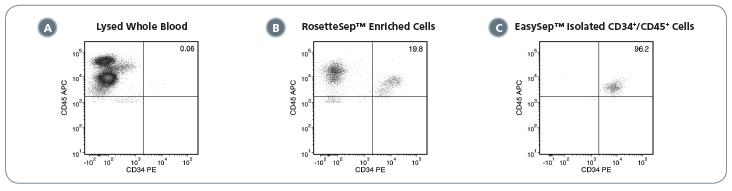
Figure 2. Typical Performance of the EasySep™ Complete Kit for Human Whole Blood CD34+ Cells
(A) In whole PB, the proportion of CD34+/CD45+ cells is typically very low (< 1%). (B) In the RosetteSep™ enrichment step, unwanted cells are removed during density gradient centrifugation. This step enriches the population of CD34+/CD45+ cells (19.8% in this example). (C) After positive selection of CD34+ cells using EasySep™ , a highly pure population of CD34+/CD45+ cells (96.2% in this example) can be obtained. Values are reported as a percentage of viable CD45+ cells.
Expansion of HSPCs in Culture
Hematopoietic stem and progenitor cells (HSPCs), including erythroid progenitors and CD34+ cells, are excellent targets for reprogramming. However, due to the low frequency of these cells in PB (typically < 1% of PBMCs) and their low reprogramming efficiency, it is necessary to expand HSPCs in culture to obtain enough target cells for reprogramming.
Our serum-free expansion media, StemSpan™ SFEM II, is an enhanced version of StemSpan™ SFEM. StemSpan™ SFEM II can be supplemented with our standardized cytokine cocktails, such as the StemSpan™ CD34+ or the Erythroid Expansion Supplements for HSPC culture. StemSpan™ SFEM II is the preferred medium for CD34+ and erythroid expansion cultures as it supports higher expansion of these cells than all leading serum-free hematopoietic media tested.
After culturing PB-isolated CD34+ cells for 7 days in StemSpan™ SFEM II and CD34+ Expansion Supplement, cell numbers can be expected to increase 20 to 50-fold with approximately 60% of the cells retaining expression of the CD34 antigen (Table 1).
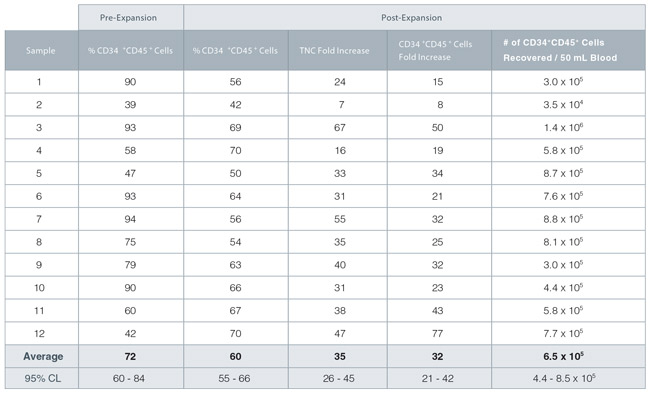
Table 1. StemSpan™ SFEM II and CD34+ Expansion Supplement Support Expansion of CD34+ Cells
TNC: Total Nucleated Cell
Our StemSpan™ Erythroid Expansion Supplement selectively expands erythroid progenitor cells, which are identified by expression of the transferrin receptor (CD71) and Glycophorin-A (GlyA). CD71+GlyA+ cells make up the majority of expanded erythroid cells and consist mostly of (basophilic) erythroblasts. A smaller population of CD71+GlyA-/low cells that contains pro-erythroblasts and more immature progenitors is also present in these cultures (Figure 3).
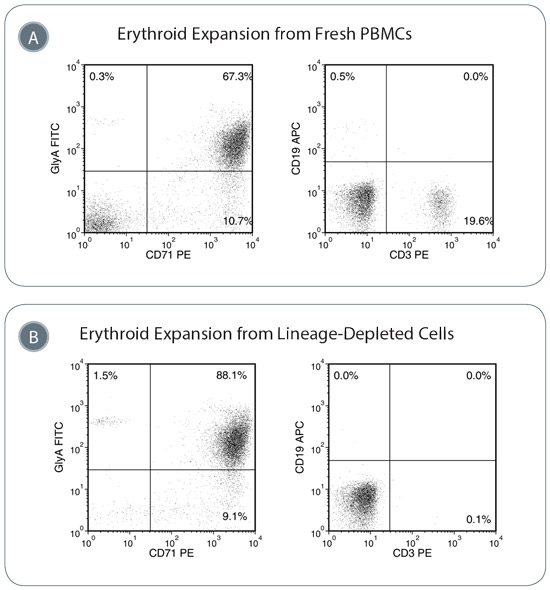
Figure 3. Erythroid Progenitor Cells are Expanded in StemSpan™ SFEM II Containing Erythroid Expansion Supplement
(A) Isolated PBMCs were expanded for seven days and then examined by flow cytometry for erythroid progenitor cells, T-cells and B-cells. Representative plots illustrate that erythroid progenitor cells (GlyA+CD71+) are enriched after seven days, though some T-cells (CD3+) and B-cells (CD19+) remain. (B) Use of the RosetteSep™ cocktail to deplete mature blood cells leads to increased purity of the expanded erythroid progenitors and negligible contamination with lymphoid cells.
Note: same donor sample used for A and B.
Erythroid expansion cultures can be initiated with PBMCs. However, mature T cells are abundant in this cell fraction and form a major contaminating cell type when culturing and expanding erythroid progenitor cells. To avoid contamination of reprogramming cultures with T- and B-cells, erythroid cultures can be initiated with blood cells that have been depleted of these mature cells (with RosetteSep™) beforehand.
Defined Conditions for Reprogramming Blood-Derived Cells
Partially reprogrammed colonies have often been observed during reprogramming. These cells are phenotypically diverse, but often fail tests of pluripotency.(12) Under feeder-free conditions, overall reprogramming efficiency is lower, though the only types of cells that emerge are fully reprogrammed,(13) indicating the importance of defined culture conditions in the process of reprogramming.
ReproTeSR™ is a complete, defined, xeno-free and feeder-free reprogramming medium optimized for the generation of iPS cells from erythroid or CD34+ cells expanded in vitro from PB. This reprogramming medium provides recognizable iPS cell colonies with less differentiated or partially reprogrammed background cell growth.
Use ReproTeSR™ during the induction phase of reprogramming (Figure 4) to obtain more iPS cell colonies than with traditional KOSR-containing human ES cell medium (Figure 5). iPS cell colonies generated with ReproTeSR™ express undifferentiated cell markers and exhibit more defined borders, compact morphology, and reduced differentiation (Figure 6).
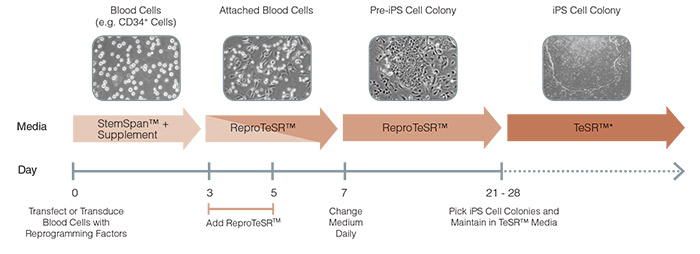
Figure 4. Schematic of ReproTeSR™ Reprogramming Timeline
ReproTeSR™ is used during the entire induction phase of reprogramming (day 3 to 21). On days 3 and 5, ReproTeSR™ is added to StemSpan™ growth media (in a fed-batch manner) to facilitate attachment of transfected cells. Attached cells are further cultured in ReproTeSR™ with daily full media changes until putative iPS cell colonies emerge (days 21-28). iPS cell colonies can then be isolated and propagated in TeSR™ media (mTeSR™1, TeSR™2, TeSR™-E8™).
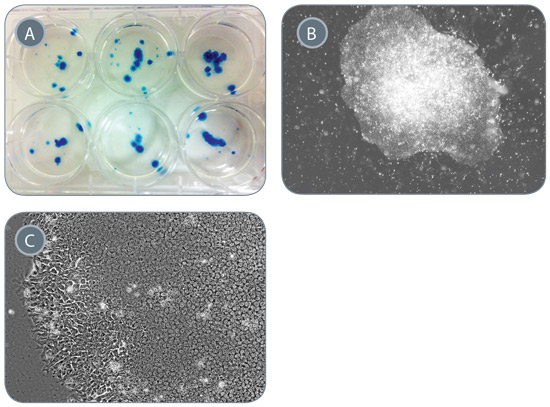
Figure 5. Generation of iPS Cells from 1 mL of Peripheral Blood
Starting from 1 mL of PB, PBMCs were enriched, and erythroid progenitors were expanded and reprogrammed in ReproTeSR™. (A) Approximately 75 iPS cell-like colonies that were positive for alkaline phosphatase expression (blue) were generated. (B-C) iPS cell colonies exhibit compact ES cell-like morphology with defined borders and high nuclear-to-cytoplasmic ratio. Representative images of generated iPS cell colonies taken at 20X (B) and 400X (C) magnification are shown.
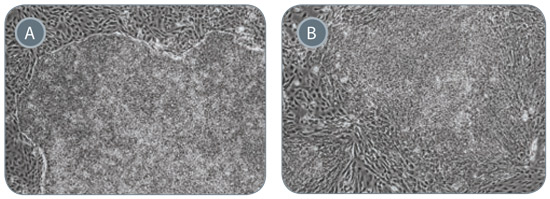
Figure 6. ReproTeSR™ Generates iPS Cell Colonies With Superior Colony Morphology
Representative images of iPS cell colonies generated from isolated CD34+ progenitor cells using (A) ReproTeSR™ and (B) human ES cell medium. iPS cell colonies produced using ReproTeSR™ exhibit more defined borders, compact morphology and reduced differentiation compared with hES cell medium. 200X magnification.
Summary
iPS cells have many applications in research, drug development, and disease management. The Erythroid Progenitor and CD34+ Progenitor Reprogramming Kits, featuring ReproTeSR™, allow you to easily isolate, expand and efficiently reprogram blood cells. iPS cells generated from this workflow can transition seamlessly to our TeSR™ family of iPS cell maintenance media and the STEMdiff™ suite of products for directed differentiation.
For more information on these and other products for blood cell reprogramming and iPS cell research, click here.
References
- Takahashi K, et al. Cell 131(5): 861–72, 2007
- Yu J, et al. Science 318(5858): 1917–20, 2007
- Aasen T, et al. Nat Biotechnol 26(11): 1276–84, 2008
- Niibe K, et al. PLoS One 6(3): e17610, 2011
- Loh Y-H, et al. Cell Stem Cell 7(1): 15–9, 2010
- Choi SM, et al. Blood 118(7): 1801–5, 2011
- Mack AA, et al. PLoS One 6(11):e27956, 2011
- Zhou T, et al. J Am Soc Nephrol 22(7): 1221–8, 2011
- Zhang SC, et al. Nat Biotechnol 19(12): 1129–33, 2001
- Nishimura T, et al. Cell Stem Cell 12(1): 114–26, 2013
- Chou BK, et al. Cell Res 21(3):518-29, 2011
- Xu H, et al. Cell Res 22(1): 142–54, 2012
- Chan EM, et al. Nat Biotechnol 27(11): 1033–7, 2009