Robust, Defined, Animal-Free Virus Production Medium Optimized for Microcarrier Culture
Steve Pettit, Grishma Patel, Mark Szczypka, Claire Y.-H Huang, Bart Tarbet
Abstract:
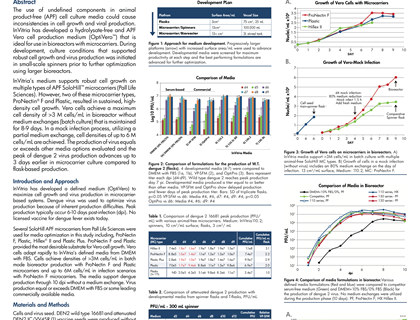
The use of undefined components in animal product-free (APF) cell culture media could cause inconsistencies in cell growth and viral production. InVitria has developed a hydrolysate-free and APF Vero cell production medium (OptiVero™) that is ideal for use in bioreactors with microcarriers. During development, culture conditions that supported robust cell growth and virus production was initiated in small-scale spinners prior to further optimization using larger bioreactors.
InVitria’s medium supports robust cell growth on multiple types of APF SoloHill™ microcarriers (Pall Life Sciences). However, two of these microcarrier types, ProNectin® F and Plastic, resulted in sustained, high-density cell growth. Vero cells achieve a maximum cell density of >3 M cells/mL in bioreactor without medium exchanges (batch culture) that is maintained for 8-9 days. In a mock infection process, utilizing a partial medium exchange, cell densities of up to 6 M cells/mL are achieved. The production of virus equals or exceeds other media options evaluated and the peak of dengue 2 virus production advances up to 3 days earlier in microcarrier culture compared to flask-based production.
[button color=”green” link=”https://www.invitria.com/wp-content/uploads/sites/2/2020/05/InVitria-Production-Medium-Microcarrier-Culture-WVC-4-2015.pdf”]View Poster[/button]