
Real Time IgG Titer Measurement Enables PAT (Process Analytical Technology) Implementation
We recently finished our Ask the Expert discussion on “Measuring IgG Titer: Need for Real-Time Results, Customization and High Comparability to HPLC.” During this Ask the Expert session, we discussed strategies for at line IgG titer measurement, comparison of this method with HPLC, customizing the IgG assay and at-line testing for perfusion culture. Other topics included the ability to measure multiple parameters and operation details.
Implementation of PAT
The successful implementation of PAT (process analytical technology) involves analysis and monitoring of key attributes within the bioprocess including measuring IgG titer. When measuring critical process parameters, such as IgG titer measurement, the need for high quality data AND real-time access are very important.
While HPLC is the gold standard, is it accessible at all stages (early cell line development to manufacturing) and at the frequency desired?
What is the impact of getting IgG titer in minutes versus days, without preparing and sending samples for HPLC analysis?
IgG Titer Measurement:
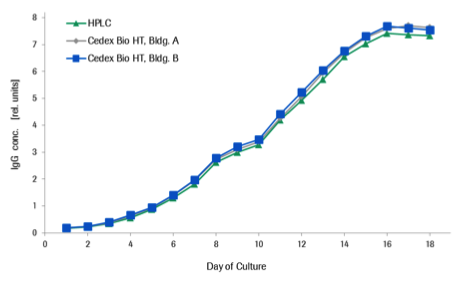
Roche Custom Biotech offers an IgG assay available on the Cedex Bio and Cedex Bio HT Analyzers which provides fast and accurate human IgG titer measurement in cell culture and fermentation media and delivers results that are highly comparable to HPLC (see Graph 1). The assay is based on a turbidimetric method using antibodies directed against the common human Fc region. Additionally, the assay calibration can be adapted specifically to the individual IgG molecule, providing optimal accuracy for artificially engineered antibodies with uncommon structures as well. When compared to gold standard methods like HPLC, the data shows close correlation with decreased turnaround times and less need for manual intervention.
The Cedex Bio and Cedex Bio HT Analyzers provide access to 21 assays including Glucose, Lactate, Ammonia, Glutamate, Glutamine, LDH, IgG, Iron, Acetate, Phosphate, Optical Density, etc, and a test menu that has continuously expanded according to the needs of the market. The systems offer on-board dilution, minimal hands-on maintenance, low sample volume requirement and wide measuring ranges. The Cedex Bio Analyzer is appropriate for low to medium sample throughput while the Cedex Bio HT Analyzer is suited for higher throughput holding up to 90 samples at once (with continuous loading), and offering advanced workflow integration capabilities.
In this Ask the Expert session, Dr. Christian Weilke, Ph.D., International Product Manager at Roche Custom Biotech, answered questions regarding the IgG assay and the Cedex Bio and Bio HT Analyzers. Christian holds a Ph.D. in chemistry and joined Roche in 1999. Since 2010, he has been working in Custom Biotech holding several different positions, and is currently responsible for the Pharma In-process Control portfolio.
Below is a sneak peek of the discussion, for a full transcript, please see – Ask the Expert – Measuring IgG Titer: Need for Real-Time Results, Customization and High Comparability to HPLC.
Question:
How do you customize this assay for different IgG molecules?
The Answer:
The Roche IgG Assay can be adapted to specific human IgG molecule(s) by a product-specific calibration. We provide a protocol where the Roche calibrator can be replaced by a sample of your individual IgG molecule with a known concentration, and the rest of the assay reagents are fully compatible, with no additional materials or hardware needed. Both the Cedex Bio Analyzers are capable of storing multiple custom calibration curves, allowing flexibility to expand the customization to obtain titers for multiple products.
Question:
We are running perfusion culture so our runs are over a month long. Can the inline sampling be adapted for that kind of timeframe?
The Answer:
“At-line testing” using the Cedex analyzers requires you to obtain a small amount of sample (minimum of about 100 µl) from the culturing system for testing. The big advantage in comparison to an integrated in-line system is that an alteration or failure of an internal sensor in the bioreactor would be fatal because process control is no longer possible (severe process risk). On the other hand, the at-line instrument can be easily maintained and you can rely on continuous process control with high data quality, and the instrument has the flexibility to be placed in close vicinity to the culture or in a dedicated lab, whatever fits better in the individual environment.
Question:
With the Cedex Bio or Bio HT how often can I take samples/measurements?
The Answer:
Both the Cedex Bio and Bio HT offer continuous sample loading and can run 1 assay, such as IgG, or any combination of the 21 available assays. The Cedex Bio HT is ideal for use in labs requiring high throughput; it holds up to 90 samples at once, with continuous loading. The system can operate un-interrupted, and as an example, can process up to 320 IgG samples per hour. The Bio is better suited for a lower throughput application; it holds 8 samples at once, with continuous loading, and can provide 60 results per batch.