Predicting Differentiation and Characterizing Pluripotent Stem Cells Using Non-invasive Multi-analyte Luminex® Assays
As part of our continuing coverage of ISSCR 2016, we will be highlighting some of the posters presented at the conference. One poster that I thought provided an innovative solution for stem cell characterization during differentiation was presented by R&D Systems, “Predicting Successful Pluripotent Stem Cell Differentiation Using Non-invasive Multi-analyte Luminex® Assays.” In the poster, R&D Systems describes their non-invasive method for predicting differentiation and stem cell characterization.
Introduction
One of the challenges facing Stem Cell Research and Cell Therapy applications today is characterizing and investigation of stem cell and differentiated populations. There are several options currently available for this type of investigation, for example, rt-PCR, western blot, immunochemistry, and flow cytometry. However all these methods are invasive, which means that valuable cells are lost during the process of characterization.
Non-invasive multi-analyte cell analysis
To address these limitations, R&D Systems has developed a non-invasive method for cell characterization during differentiation. This method utilizes the flexibility of Proteome Profiler™ Antibody Arrays and Luminex® Assays to assess the expression of multiple analytes in cell culture supernates. Because this method uses cell culture supernates it is non-invasive and enables the detection of changes in cytokine expression throughout the progression of stem cell differentiation.
Poster Highlights
To demonstrate the effectiveness of this method, R&D Systems conducted a study, presented in the poster, that evaluated the multi-analyte screening capabilities of Luminex assays to profile the levels of cytokines and growth factors in cell culture media at key stages during the differentiation of pluripotent stem cells into hepatocytes. Cytokine and growth factor expression profiles were obtained from human induced pluripotent stem (hiPS) and human embryonic stem (hES) cell lines with known differences in hepatocyte differentiation efficiency. Because analytic samples are obtained from culture media, the cells are able to continue through the differentiation process and be analyzed for efficiency by assessing albumin expression, a marker of functional hepatocytes.
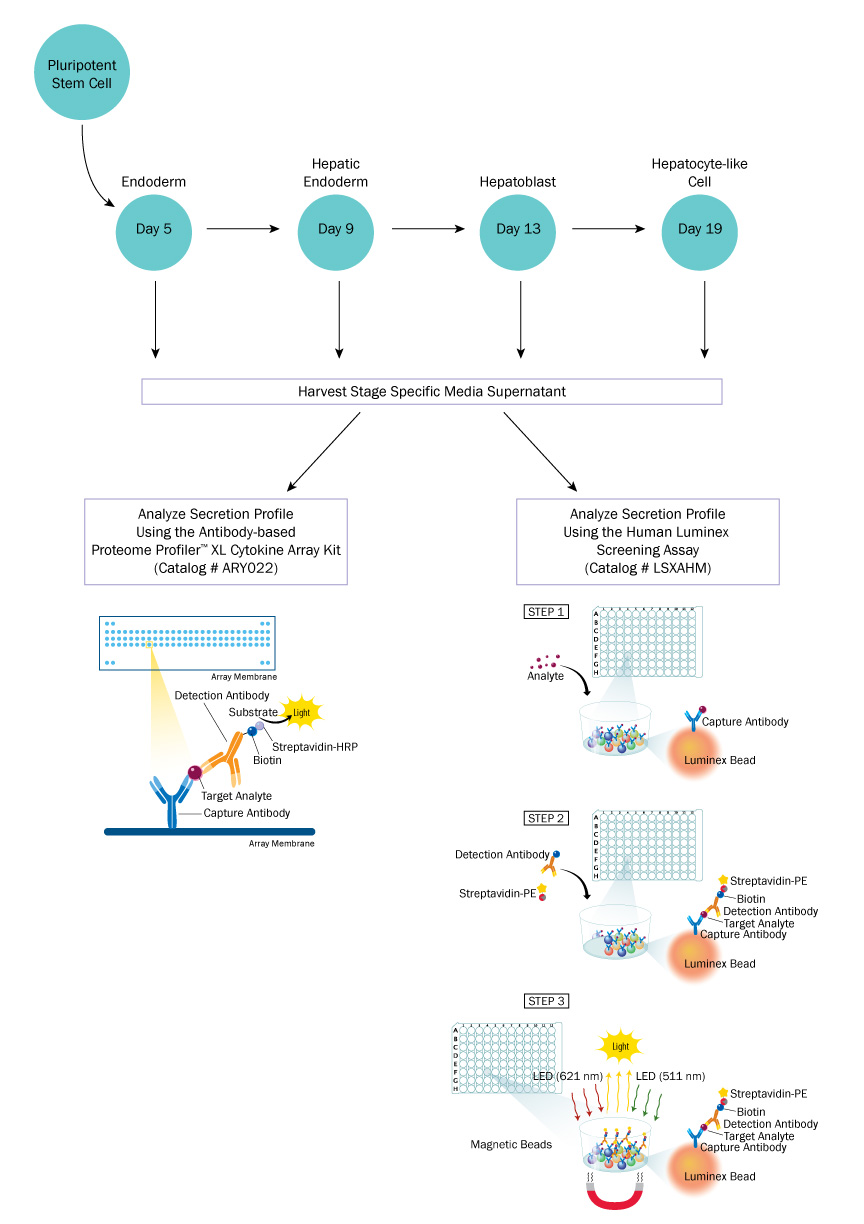
By analyzing the multi-analyte profile of cell lines with robust differentiation efficiency and comparing them with cell lines with lower efficiencies, the goal would be to identify particular analytes that are indicative of differentiation success and could be used in predicting differentiation. This data could also be used to identify any other factors that would enhance either differentiation or maturation of differentiated cells. A diagram of the study outline is shown below (Figure 1).
Figure 1: Experimental Study Outline
About Luminex® Assays
Luminex Assays utilize color-coded polystyrene or superparamagnetic beads coated with analyte-specific antibodies. Beads recognizing different target analytes are mixed together and incubated with the sample. Captured analytes are subsequently detected using a cocktail of biotinylated detection antibodies and a streptavidin-phycoerythrin conjugate. It functions similar to an array, but with the Luminex you have the ability to look at specific analytes.
Results
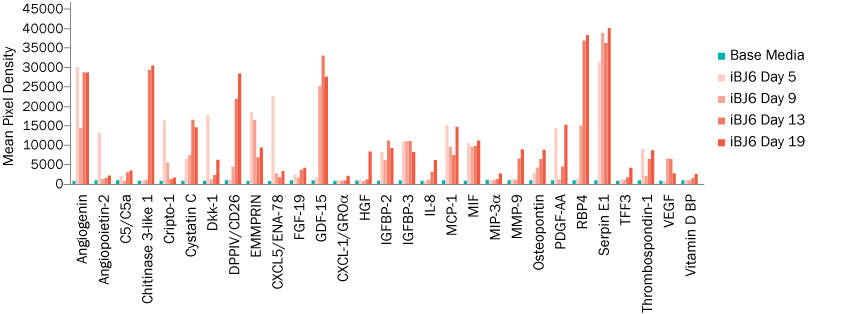
R&D Systems was able to demonstrate that, using cell culture supernates, the Proteome Profiler and Luminex multi-analyte assays were able to detect changes in cytokine expression throughout the progression of stem cell differentiation into hepatocyte-like cells. As such, the secretion profiles provided a non-invasive method for the characterization of differentiation (Figure 2). These signature differences may provide a method for predicting differentiation success and cell line specific optimization of differentiation protocols.
Figure 2: Sample Data from Luminex®
Other benefits of using this method include:
- Up to 100 biomarkers can be simultaneously profiled
- Cost-effective way to profile multiple markers
- Assays can be performed in 3-3.5 hrs
- Assays require a small sample volume (<50 µL)
- No harvesting of cells required, permits analysis of end cell population
- Enables an early assessment on whether differentiation is going well
For more data and full details of the highlights discussed here, please click on the poster below to view in full size.
For more information on this technology, please view Dr. Aho’s webinar “How to Non-invasively Monitor Stem Cell Differentiation using Luminex Technology,” now available on demand.
As Stem Cell Research moves closer toward therapeutic application, there is a recognized need to increase the quality and consistency of pluripotent stem cells and their derivatives. While variability in the efficiency of stem cell differentiation can undermine data interpretation and slow research progress, the ability to quickly monitor changes in cell phenotype without sacrificing yield continues to be a challenge. In this webinar, Richard Feurstenberg and Joy Aho discuss Luminex technology, its power as a non-invasive tool for monitoring stem cell differentiation, and its utility for troubleshooting and optimizing stem cell culturing and differentiation conditions.
To learn more:
- Luminex Assays: https://www.rndsystems.com/products/luminex-assays
- Guide to Choosing an Immunoassay: https://www.rndsystems.com/products-highlights/choose-right-immunoassay-get-your-answer
- Stem Cell Products: https://www.rndsystems.com/products/stem-cell-products