
Bioengineered Organ Implants Poised to Change Treatment Paradigms in Esophageal Cancer
Podcast: Download (Duration: 15:59 — 22.0MB)
Subscribe Here: Apple Podcasts | Spotify | RSS | More
Subscribe to the Cell Culture Dish Podcast on: iTunes | Google Play
Show Notes:
We began the interview talking with Dr. LaFrancesca about how Biostage is using their Cellframe technology to improve on the current standard of care for esophageal diseases. Dr. LaFrancesca began by explaining that esophageal disease patients are divided into two patient populations – adults and pediatric. For adults affected by esophageal disease or cancer the standard of care is to pull up their stomach to meet the remaining portion of esophagus after resection or to remove a section of the intestine and use it to replace the removed portion. These surgical procedures are very complicated and involve surgeries into multiple body cavities. The mortality and morbidity rate for these treatments is very high and have serious associated complications. In most pediatric cases the babies are born without an esophagus and in this case there is no standard of care. The surgeon will utilize the same techniques used in adult cases, but there are serious consequences for the infants including failure to thrive and issues with growth.
Dr. LaFrancesa stressed that this is a pressing problem in healthcare due to the lack of good treatment options. In addition to the dangers that these surgeries present, there is also a serious negative impact to the patient’s quality of life, even when the procedures are successful.
Next I asked Dr. LaFrancesca about the alternative therapy that Biostage is working on. He explained that the Cellframe technology employs a multi-step process in which the patient’s own stem cells are taken from a simple adipose/fat tissue biopsy, expanded and banked, and then seeded onto a proprietary scaffold that mimics the natural dimensions of the organ being regenerated. After several days in a rotating bioreactor, the stem cell-containing scaffold is ready to be implanted. Cellframe technology is designed to deliver the necessary cues for triggering, guiding and modulating the regenerative process. Below please see a visual representation of how Cellframe technology works:
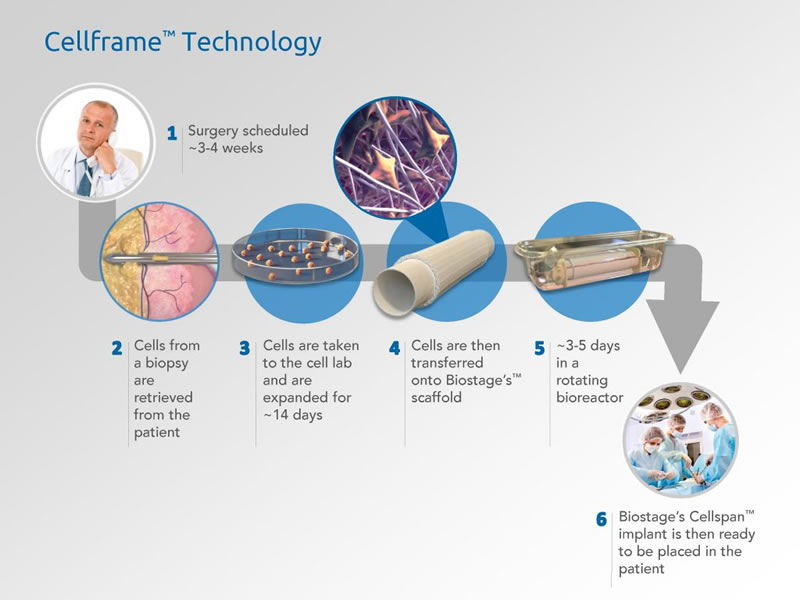
Currently, the therapeutic is in preclinical safety studies required prior to beginning clinical trials in humans.
Next I asked Dr. LaFrancesca about challenges in R&D that they have overcome so far. He said that they have overcome many challenges, first just in developing a product that is biocompatible and can harness the bodies’ own healing ability. Now that they have accomplished this, they are very excited about the very relevant animal models that they have created. They have demonstrated that these models are both robust and reliable. The study results have been very positive.
Next I asked Mr. McGorry about Biostage’s successes to date. He explained that they have dedicated many years to the development of the product with the help of collaborators including the Mayo Clinic and Connecticut Children’s Hospital. In late 2016, they received orphan drug designation from the FDA for treatment of adults and children with esophageal disease. They also continue to generate several peer reviewed articles with more to come in 2017.
Last I asked Mr. McGorry about upcoming milestones and he said that they will continue to move the product toward clinical trials with a goal of filing their IND in 3rd quarter 2017. Of course the ultimate goal is to move the product to FDA approval and commercialization.
Mr. McGorry ended the interview by thanking their collaborators.
Cellframe technology is based on the concept of in situ tissue regeneration using the body’s own biologic resources and reparative capability in combination with tissue-specific biomaterials implanted at the sites of disease or injury.
Cellframe is a newly developed technology that harnesses the full potential of the in vivo microenvironment to achieve complete tissue regeneration.
Biostage is investigating its Cellframe technology to replace diseased sections of the esophagus, trachea and bronchus, possible life-threatening conditions that are underserved by current therapies.
For more information, please watch this animation to learn more about how Cellframe technology works.