
Animal Component Free Recombinant Leukemia Inhibitory Factor Critical for Enhancing Human Neural Stem Cell Proliferation
A Guest Blog by Randall Alfano, Ph.D., Cell Culture Scientist, InVitria
Stem cell-based therapy has the potential to treat an array of human diseases. However, to realize the full therapeutic potential of these cells, a cost-effective cell culture medium is needed that is free of human or bovine-derived serum proteins. Thus, cost-effective and animal component-free recombinant proteins and cytokines are needed to produce such mediums. One such cytokine, Leukemia Inhibitory Factor (LIF), has been shown to be a critical paracrine factor that maintains stem cell pluripotency in murine embryonic stem cells and human naïve stem cells while simultaneously inhibiting differentiation. We recently produced a recombinant human LIF (rhLIF) in a plant-based protein expression system known as ExpressTec (Youngblood, et al., J. Biotechnol., 172, 67-72, 2014). Recombinant proteins derived from ExpressTec are cost-effective at large scale and offer a completely animal-free and low endotoxin source of recombinant proteins for cell culture applications. A previous study described the cloning and expression of the ExpressTec-made rhLIF and further demonstrated that the biological activity of the rhLIF protein was equivalent to a commercially-available rhLIF protein in the C57BL/6 N Lex3.13 mouse embryonic stem cell system with significantly less contaminating endotoxin (Table 1).
| Vendor | Source | Endotoxin Specification (EU/ug) |
|---|---|---|
| Santa Cruz Biotechnology | E. Coli | Not Specified |
| Millipore | E. Coli | < 1 |
| Life Technologies | E. Coli | < 1 |
| ORF Genetics | Barley Grain | < .05 |
| Sigma-Aldrich | E. Coli | < 1 |
| InVitria (b) | Rice Grain | < .002 |
b. Endotoxin level was measured using a Pyrogene Endotoxin Assay
We have further expanded this research to include human stem cells by demonstrating that the rhLIF protein enhances human neural stem cell proliferation in vitro. We exposed H9 embryonic stem cell (ESC)-derived neural stem cells (hESCD-NSCs) to 10 ng/mL LIF for 96 hours in order to compare rhLIF to a commercial human LIF standard produced in E. coli. When cultured in DMEM/F12 supplemented with 2 mM Glutamax, 1% N-2, 10 ng/mL bFGF and EGF, hESCD-NSCs exhibited a doubling time of 58.97 ± 4.43 hours. However, addition of human LIF, produced either in E. coli or with ExpressTec, exhibited a significant 41% (p = 0.0006) and 43% (p = 0.00006) reduction in doubling time to 35.63 and 34.75 hours, respectively (Figure 1).
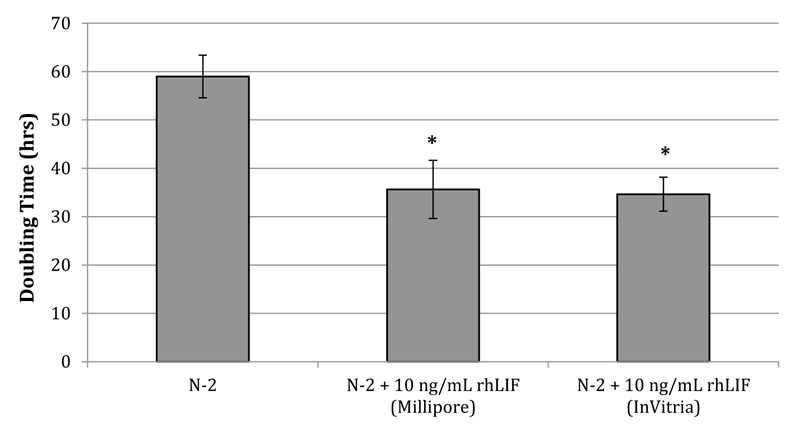
Our previous studies demonstrated that the ExpressTec-made rhLIF was found to be highly active as determined by a modified M1 cell differentiation assay and was sufficient in retaining pluripotency of C57BL/6 N Lex3.13 mouse ESCs through multiple subcultures. Here, we have expanded the application of the rhLIF to human stem cells by demonstrating that the rhLIF enhances human neural stem cell proliferation in vitro. This data indicates that the rhLIF protein is active in human stem cells and thus can be an economical growth factor option for human stem cell subtypes that are responsive to LIF supplementation. Thus, inclusion of the ExpressTec-produced rhLIF in clinical stem cell media can improve the affordability of new stem cell therapies and facilitate the translation of this promising technology to clinical practice.
For questions regarding rhLIF or any of the InVitria products, please contact our technical support.