
A Complete Media System for Transient Transfection in CHO Cells
CHO cells are considered the workhorse of the biomanufacturing industry. According to Jayapal, Wlaschin, Yap, and Hu (2007) nearly 70% of all recombinant protein therapeutic production is done using CHO cells, and pharmaceutical sales for biologics produced in CHO are over $30 billion worldwide.
CHO cells have been successful because they are a great choice for biomanufacturing. CHO cells have the ability to thrive in suspension cell culture, adapt to serum-free and chemically defined media formulations, and their capacity for incorporation of post-translational modifications necessary for effective human therapeutics provide distinct advantages over other cell lines. These advantages make CHO cells an easy choice for large-scale biomanufacturing, but until recently, CHO cells were not often used for smaller-scale drug development applications.
Transient Transfection and Protein Production in CHO Cells
Transient protein production enables quick and efficient production of milligram to gram quantities of recombinant protein, which saves time and cost compared to developing a genetically stable cell line for use in bioproduction. This is particularly important in drug development, where timelines are short and only a relatively small amount of material is required. While CHO cells were not historically used in transient transfection systems, due to low bioproduct expression levels as compared to HEK293 systems, it was widely recognized that the ability to produce transient CHO-derived proteins during drug development would be very beneficial. Using CHO cells rather than other cell lines would reduce potential changes in protein function and/or quality between early research material and clinical or commercial product. Due to recent advances in transient transfection CHO systems, protein expression levels have increased to make CHO cells an attractive option for transient protein expression.
Simplifying Transient Production in CHO Cells
Recently, Irvine Scientific launched their BalanCD® Transfectory™ CHO system of medium and supplements for transient transfection in CHO cells. The system provides a scalable platform approach to transient transfection CHO with milligram to gram-scale yields of recombinant proteins.
Key features of the BalanCD Transfectory CHO system include:
Optimized Productivity
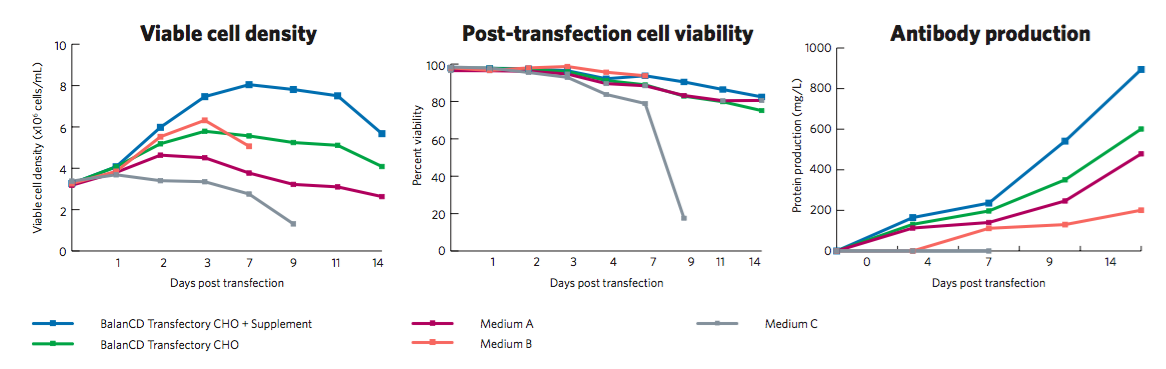
In order to meet drug development product needs and provide an advantage over HEK293 systems, consistent, high expression levels must be achieved in transient CHO. According to data presented by Irvine Scientific, the BalanCD Transfectory CHO system provided improved productivity and growth compared to other media (Figure 1).
Improved productivity and growth compared to other commercially available media
Process Consistency
The formulation of BalanCD Transfectory CHO is based on the BalanCD CHO platform of growth media and feeds. This allows for media consistency from transient production through large-scale manufacturing.
In addition, there is no need to exchange the medium before or after transfection. Since the system supports small- and large-scale transient transfection, the same medium can be used before and after transfection.
Flexibility
The BalanCD Transfectory CHO System is chemically-defined (hydrolysate-free) and animal component free. It provides a single medium for cell growth, PEI-DNA complex formation and transfection. As part of the system, there is also Transfectory™ Supplement, which is an optional hydrolysate supplement that maximizes post-transfection cell growth and protein production. Additionally, Anti-Clumping Supplement is also included as part of the system, to reduce cell aggregation in suspension culture.
For more product information, please visit BalanCD Transfectory CHO System.
To obtain samples, please contact Irvine Scientific:
Tel: +1 949 261 7800
Email: getinfo@irvinesci.com