
Solve Production Challenges of Difficult to Express Proteins with Scalable, Continuous Manufacturing
At this year’s Boston Biotech Week there were many interesting talks on continuous biomanufacturing and associated new technologies. In particular, there was a great deal of discussion around how to handle difficult to produce proteins. These manufacturing challenges included proteins that were difficult to express and/or were unstable. One very interesting talk, given by Scott Waniger, Vice President, Bioservices Division, Cell Culture Company, was titled “Solving Production Challenges of Difficult-to-Express Proteins with a Scalable, Continuous Manufacturing Bioreactor: A Case Study” and focused on the use of perfusion bioreactors (hollow fiber) to create both continuous upstream production and address the issue of cost effective manufacturing of difficult to express proteins.
Mr. Waniger began the talk by describing several of the protein production challenges in the market.
They included:
- The need for new classes of engineered proteins (not human IgG’s) which typically require smaller commercial quantities for orphan/rare disease and smaller dosages due to better drug targeting, in-vivo imaging molecules and more potent biologics (i.e. ADC’s, bi- and tri-specific’s).
- CHO and HEK-293 sometimes just cannot make these proteins at high titer levels at multiple g/L titers no matter how much genetic engineering is done.
- The Precision Medicine Initiative (Obama administration January 20th, 2015) is an emerging approach for disease treatment and prevention that takes into account individual variability in genes, environment, and lifestyle for each person.
- Current platforms for manufacturing are geared towards production of hundreds of kilograms of protein so making small commercial (sub kilo-gram) GMP batches are not cost effective.
- Biosimilar’s will allow multiple drug companies to make the same drug. This will mean smaller batches but will also help to bring costs down through competition.
- Smaller markets (South America, Africa, Middle East) will require government operated manufacturing within their country to supply their smaller populations with biologics.
- Some protein expression systems do not make high quality protein in suspension and traditional fed batch tank upstream bioreactors due to shear, high osmolarity, low pH, heterogeneous metabolic concentrations causing change in glycosylation, excessive proteases etc.
The conclusion is that there is a need for small scale (< 1kg/year) GMP manufacturing. At this scale, it makes sense to think about a commercial alternative to traditional batch/fed-batch systems.
Mr. Waniger went on to describe possible alternatives to the traditional batch/fed-batch system, including continuous manufacturing. The case studies presented provided examples of how low expressing proteins could still be commercially feasible in an alternative manufacturing system.
Case study 1: Scalable technology that reduces scale up validation studies
In the first case study they examined five different IgG producing cell lines and the ability to scale up manufacturing while reducing the need for validation studies. They normalized the data for the amount of protein manufactured per hollow fiber cartridge. They were able to demonstrate scalability for a 10-fold expansion of bioreactor capacity. The cell substrate (cartridge) space did not change in form, fit, size or function. Scale up was achieved by simply connecting multiple cartridges in parallel (Figure 1).
Figure 1:
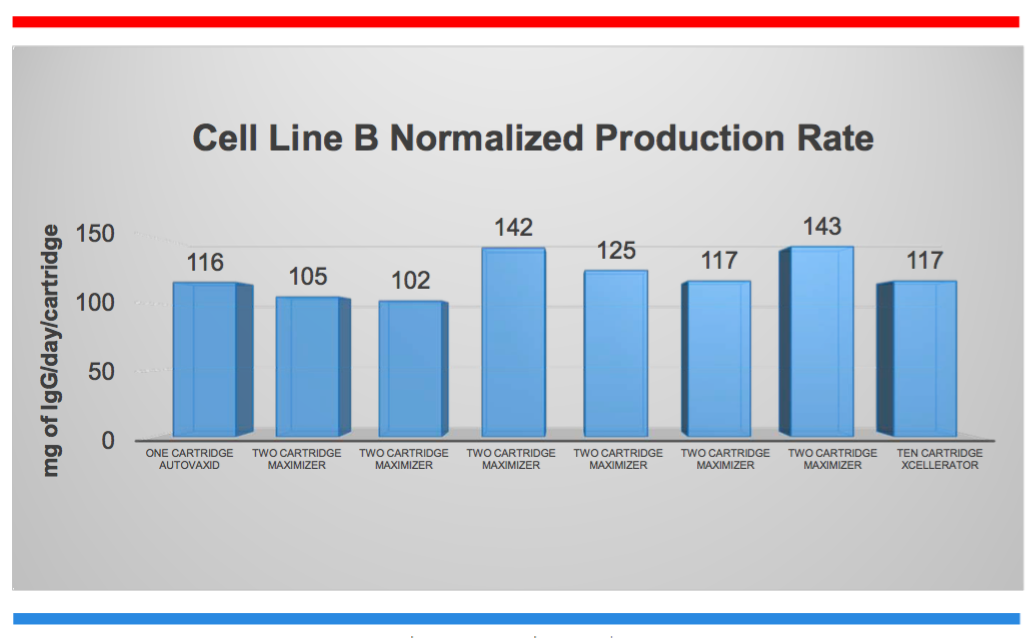
Case Study 2: Continuous Protein Expression Manufacturing
In the second case study, Mr. Waniger presented a case where a TNF Beta fusion protein was expressed at (0.23 mg/L) in shake flasks, but this titer was too low. The project was then moved to a 70 day perfusion culture in a hollow fiber bioreactor and this resulted in 54 mg of protein in 5 L of supernatant. Since the protein was large and was concentrated in the cell compartment of the cartridge, more protein could be produced in lower overall processing volumes than would be possible if a stirred tank bioreactor was used (Figure 2).
Figure 2:
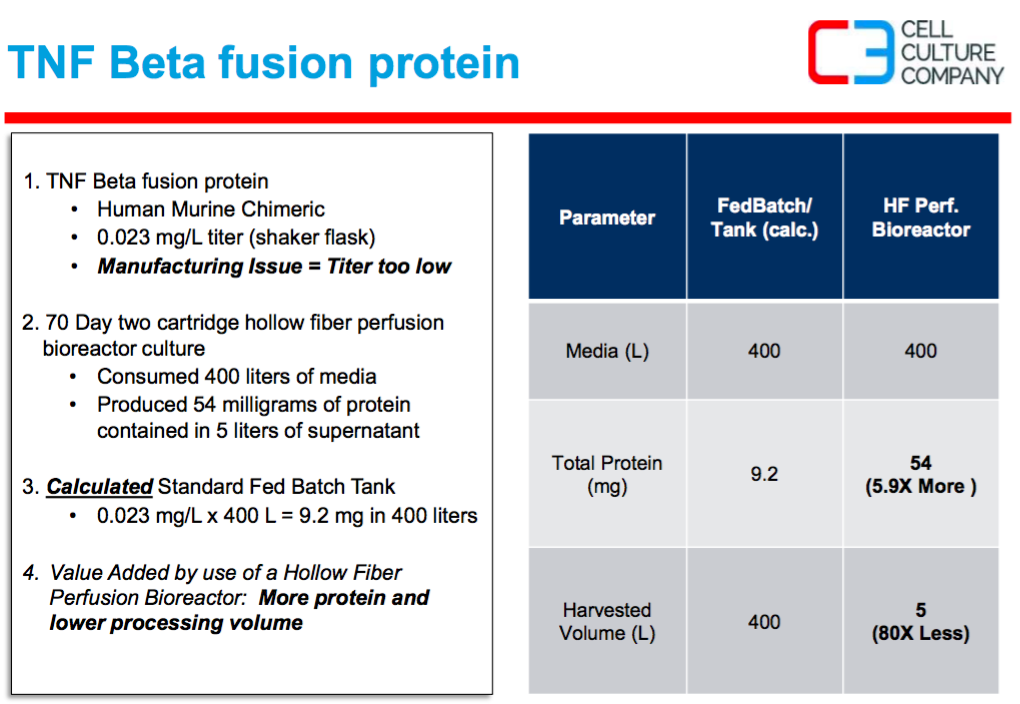
Mr. Waniger described that the more in-vivo like cell environment found in the hollow fiber culture supported the cells at ~109 cells per mL of space. The more in-vivo like environment also supports steady state metabolic activity and long lasting protein expression production (Figure 3).
Figure 3:
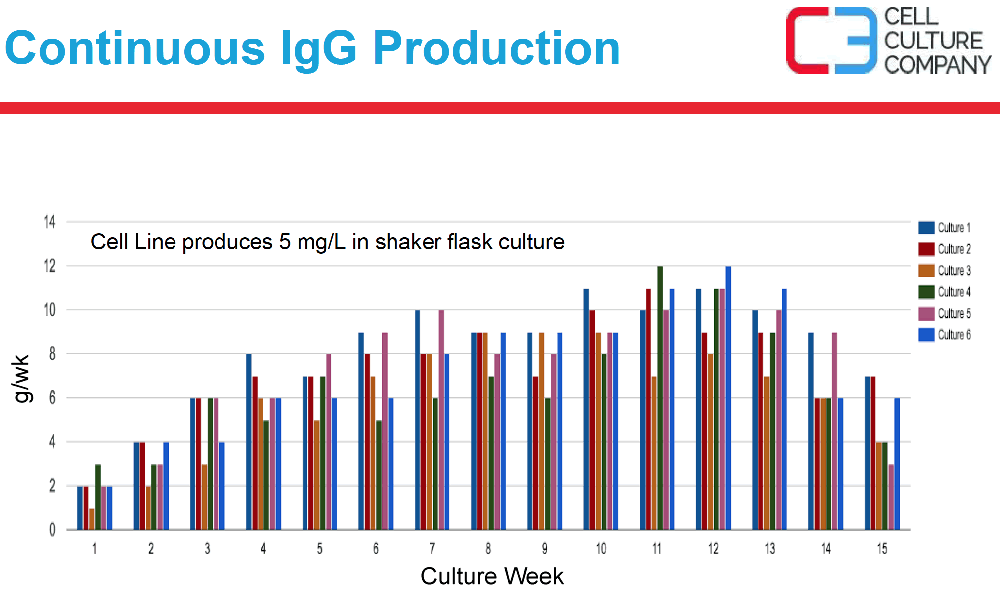
Case study 3: Difficult-to-Express Protein (< 10 mg/L titers) Manufacturing
The last case study discussed a recombinant protein expressed at 0.012 mg/L in shake flasks, again this titer was too low. In a perfusion hollow fiber system 220 mg of protein was produced and contained in 15L supernatant. (Figure 4). The advantage of perfusion was less raw material consumption and smaller downstream processing volumes than would be possible using a stirred tank bioreactor.
Figure 4:
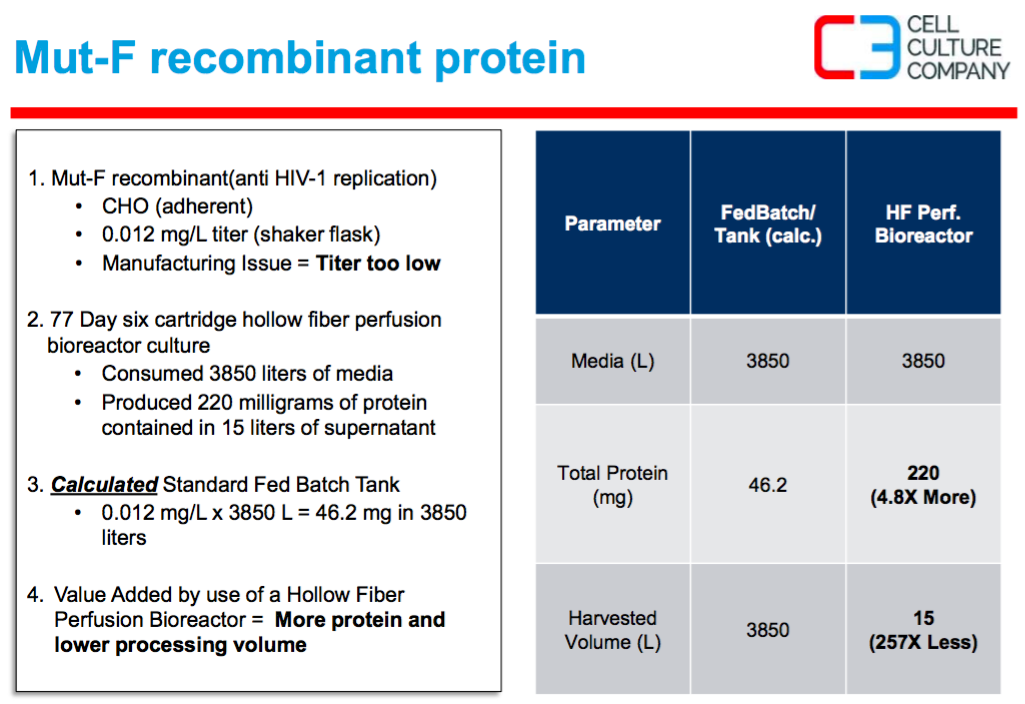
Having an alternative manufacturing system is important, for example a CHO cell line with expression of only 5 mg/L would normally be considered too low for commercialization. However with the higher cell density and longer runs that perfusion provides, this could become cost effective without the need to re-engineer the cell line or eliminate the project entirely. In this instance an alternative manufacturing system could rescue a project, especially if the product is needed in low volumes.
Summary
In summary, Mr. Waniger concluded that hollow fiber bioreactors allow companies to manufacture more protein in a smaller final volume, which provides a much simpler and more cost effective downstream purification process. In the hollow fiber bioreactor perfusion process, material is already concentrated and cell debris free and ready to purify after harvest, thus eliminating the clarification step.
In addition the high cell density in smaller volume, allows for a single-use, smaller manufacturing footprint, thereby enabling a more flexible facility with easy scalability. Mr. Waniger said that commercial scale manufacturing is about the size of a home refrigerator, this provided the perfect visualization for how small the manufacturing footprint really is.
Lastly, the system permits cell runs for months at a time with very little support needed. The hollow fiber bioreactors are designed for long-term use with little downtime. This frees up operator resources for other projects and reduces potential for human error. As an example, bioreactor set up time is about two hours, seed train preparation is 6 hours providing a very short timeline to full operation. Scale up of the cells in a traditional seed train actually takes place inside the cartridge; there is no traditional seed train with culture transfer and larger vessels. A 1600 liter equivalent hollow fiber bioreactor system can be inoculated with as little as 2 liters of cell culture volume or about 2 x 109 total viable cells. This saves set up time, effort, footprint and cost.
Conclusions
The evolving biomanufacturing landscape has created an opening for manufacturing processes that are more scalable and flexible with reduced capital investment and operating costs. In exploring hollow fiber bioreactors for a previous article “Hollow Fiber Provides a Sweet Spot for Several Biomanufacturing Applications,” I found that there are many potential applications for this technology. While in most cases the large volume manufacturing of monoclonal antibodies (hundreds of kilograms) may still be best suited for stirred tank bioreactors, there are several products and manufacturing scenarios that could benefit from the advantages, flexibility and capabilities of fully automated hollow fiber bioreactor systems.
After the talk, I was fortunate to be able to talk with Mr. Waniger and ask him some questions about hollow fiber bioreactors and perfusion culture. Here is our interview.
Question:
In your talk you discussed how hollow fiber technology could really help to solve the challenges around difficult or low expressing proteins. What is it about the hollow fiber bioreactor technology that makes it so well suited to handle these products?
Answer:
The cell substrate environment that the perfusion bioreactor provides, permits cells to be maintained in stationary phase for extended periods of time. The length of the culture is typically dependent on how much protein is needed. Not, when the cells are dead.
Question:
Another hot topic at this year’s conference was how to handle unstable proteins. Do you see a fit for hollow fiber to address some of these challenges as well?
Answer:
The perfusion rate for both nutrient delivery and product removal from the cell mass is variable and can be optimized to each and every cell line and protein. The process control allows for multiple parameters (temperature, oxygen, glucose levels, lactate levels, protein titers, etc) to be highly varied, to meet conditions for almost any mammalian or insect cell line and its resulting protein of expression.
Question:
In your case study examples, you discuss proteins that have such low expression that they might either be dropped or require a complete cell line engineering project to make them commercially feasible. Do you see hollow fiber as a way to “rescue” projects that just don’t make commercial sense but are attractive in other ways?
Answer:
The perfusion hollow fiber bioreactor systems are small and mobile and can be moved from lab to lab or suite to suite depending on the facility needs and design. Our smallest automated bioreactor system occupies approximately 6 sq ft of bench space and is equivalent to an 80 liter batch/fed-batch system. This system is linearly scalable by 20 fold. The use of this low cost pilot scale model allows operators to generate appreciable quantities of protein from their low secreting cell lines, without having to spend extensive resources on cell line development to target that holy grail of > 1 g/L.
Question:
I see another real advantage for this technology being its small footprint and I really liked your home refrigerator comparison. Can you explain for readers why a small footprint can be important?
Answer:
The small footprint and simple facility requirements (100% CO2 source, 120v power and a network connection) allows for a facility to place these closed bioreactor systems almost in any type of lab environment. They all fit through standard sized doors and can easily be moved. Our floor model is on wheels and our two bench top models are easily lifted by two adults.
Question:
Could you explain a bit more about how the seed train in hollow fiber bioreactors works?
Answer:
As the hollow fiber is a perfusion bioreactor, the delivery of nutrients to the cell mass can be adjusted to provide exactly the quantity of nutrients the cells need. Typical inoculation of a single cartridge would contain about 1 x 108 cells, in a hold up volume of 0.5 liter (inside the tubing and cartridge network). The perfusion rate can be as low as 0.5 L/day to start with and reach a maximum rate of 10 L/day. This rate is increased by monitoring the levels of metabolites such as glucose, lactate, ammonia, l-glutamine etc. As the cells consume the metabolites and produce waste, these levels will indicate to the operator what the cell needs. In simple terms, as time passes and the glucose/l-glutamine levels fall and the lactate/ammonia levels increase, the operator increases the perfusion rate of the growth media to deliver more nutrients and remove more waste. These processes are optimized for each cell line and each protein. In simple terms, the cartridge is inoculated at <1% confluent and within 10-14 days reaches 100% confluency.
Question:
Another part of your talk that really interested me was the potential benefits to downstream purification and the elimination of the clarification step. Can you explain these in more detail for readers?
Answer:
The analogy I use to answer this common question, refers to the operation of a TFF device. There is a retentate and a permeate in the cell culture device. The nutrients and waste products are perfused through the permeate side and the porous nature of the membrane allows for simple diffusion across the membrane to bathe the cells. Since the membrane pores are too small for the cells and the protein of interest to cross, the retentate side of the membrane, which is also connected to a set of perfusion pumps can be adjusted to collect an optimized quantity of media that contains the protein of interest. The tight bundle of fibers retains the vast majority (>99%) of cells yet allows the soluble protein in the media surrounding the cells to be removed. Using the 1 cartridge system as an example, each day ~ 5 liters of media is perfused in and out of the cartridge through the non-cell side of the membrane (permeate) delivering nutrients and removing waste. On the cell side of the membrane (retentate) the removal of the media containing the protein of interest can be removed at a rate of ~ 0.05-0.1 liters per day passing through a 0.2-0.45 m filter. This ratio allows for an approximately 50-100X concentration of protein that is cell debris free.
Question:
In a previous article, I talked about a sweet spot in terms of volume and application for hollow fiber bioreactor technology and you described some specific examples in your talk. Can you expand on when one would want to explore the use of this technology vs. when it would be more appropriate to use stirred tank bioreactors.
Answer:
We are currently involved with a media development program to optimize the nutrient requirements for a high expressing CHO cell line to be cultured in a hollow fiber perfusion system. We are a few months away from delivering this data to the market. In the mean time, I can tell you that for those mammalian expression systems where the quantities are sub kilogram per year, this is a platform that will be cost effective. We have been providing these bioreactor systems to the market since 1985 and we have a solid customer base that continues to make these quantities of expressed proteins. The answer to your question is that the market has told us that this is a cost effective platform for these smaller quantities of protein.
For more Boston Biotech Week Coverage, please see:
“Boston Biotech Week 2016 – Downstream Coverage – In case you missed it!”
“2016 BioProcess International Award Winners – Upstream and Analytical”
“2016 BioProcess International Award Winners – Downstream and Facilities”
Don’t miss BioProcess International West – BPI West in San Francisco February 27- March 2, 2017