
Automated Optimization of IgG Production in CHO Cells
Introduction
Monoclonal antibodies have long been an essential tool for cell and molecular assays and have moved into the clinic as part of a shift from small molecules into biologic treatments for disease. Antibodies are typically produced from a cell line that has been screened to ensure the protein is expressed with the desired post-translational modifications, target specificity and affinity, and at relatively high levels. Once this producing line has been identified, one can achieve higher titers by optimizing the culture media to increase cell growth and/or protein production.
Perhaps the most common cells for protein production are Chinese hamster ovary (CHO) cells, which can be adapted to grow in adherent or suspension cultures, the latter of which allows for more cells per unit of media and culturing in bioreactors. A number of “optimized” media for culturing CHO cells are commercially available, however, each expression clone may have different conditions that will maximize protein production such that these media may be suboptimal.
An ideal approach to media optimization is using a factorial design of experiment (DOE), where a variety of media components are tested at different concentrations in combination with one another. However, these factorial experiments rapidly increase the number of conditions that require testing. Common ways of quantifying the production of IgG or other proteins are frequently labor intensive (i.e. ELISAs) or prohibitively slow (i.e. HPLC), particularly at the high throughputs required for DOE. In contrast, the Octet platform (Pall ForteBio, Figure 1) uses Bio-Layer Interferometry to detect real-time binding of molecules as a means of quantification or kinetic analysis. This technology essentially eliminates any sample preparation beyond an optional dilution step and the high throughput models can process up to 96 samples simultaneously.

Here we show how coupling this high throughput IgG analysis with the Biomek FXP Automated Workstation (Figure 1), enabled the DOE optimization of IgG expression in a CHO cell line. The Biomek FXP Workstation was used for to prepare 96 media combinations and plate cells in replicate conditions, generate sample and reagent plates for the Octet HTX system, and initiate XTT assays to gain a better understanding of the effect of media components on CHO cell growth.
Materials and Methods
CHO Cell Culture
CHO DP-12 clone #1934 (ATCC) is a CHO line stably expressing recombinant human IgG1 antibodies against Interleukin 8. CHO cells were maintained under adherent conditions in DMEM + GlutaMAX (Thermo Fisher), 10% fetal bovine serum (Thermo Fisher), 2µg/mL recombinant human insulin (Sigma), 0.1% Trace Elements A and B (Corning), and 200nM methotrexate (Sigma). Cells were cultured at 37°C and 5% CO2.
Factorial DOE Media Optimization
Basal media for optimization experiments was CD OptiCHO (Thermo Fisher). Repeated passaging in OptiCHO caused cells to grow in suspension, however an initial plating maintained adherent characteristics. This adherence allowed image-based cell counting and prevented cell transfer during media sampling for IgG analysis.
Additional components tested for enhancement of IgG production include Polyamine, SPITE, MEM Non-essential Amino Acid Solution, Bioreactor pH Adjustment Solution (“bioreactor buffer”, all from Sigma Aldrich), GlutaMAX, HEPES, and CHO CD EfficientFeed A (all from Thermo Fisher).
We utilized a Biomek FXP Workstation featuring Span-8 pipettors and a 96-channel head with enhanced selective tip (EST) functionality to generate factorial combinations of media components (see Figure 2 for workflow). The EST feature enables any combination of tips to be loaded on the multichannel head, which aided the factorial approach when stamping sets of 4, 16, or 48 wells at once. The Biomek Workstation was contained within a HEPA-filtered enclosure to maintain the sterility of the cell cultures during manipulation. The resulting 96 conditions were plated in quadruplicate wells of a 384-well clear-bottom plate. 40,000 CHO cells were then plated in each well for a total volume of 100 µL/well. Cells were incubated at 37°C, 5% CO2 for 2-4 days prior to IgG and growth analysis. Creation of the media combinations and cell plating was completed in 20 minutes.
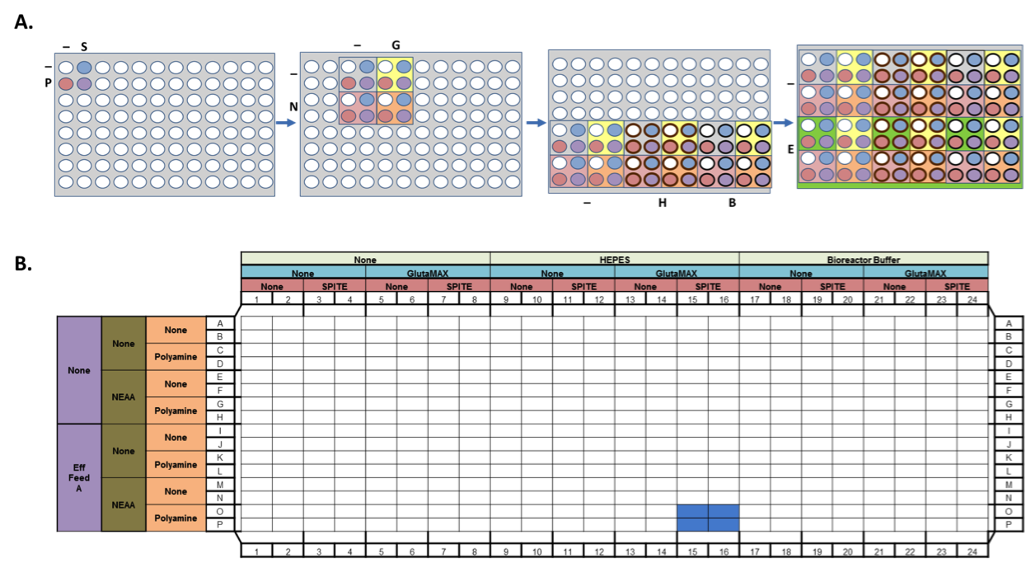
IgG Quantification
For IgG quantification, we utilized the Biomek FXP Workstation to transfer 50 µL of cell media from the culture plate to a 384-well black tilted-bottom plate (Pall ForteBio). Samples were then analyzed on an Octet HTX system using Protein A Dip and Read™ Biosensors to bind IgG directly from the media. Probes were first hydrated with basal OptiCHO media and regenerated between quadruplicate wells with 10mM glycine, pH 1.7 and neutralized with basal OptiCHO media. Hydration and regeneration/neutralization plates were also prepared by the Biomek FXP Workstation. The entire automated process was completed in under 7 min and processing of 384 samples took less than 20 minutes on the Octet HTX system when using 96 biosensors.
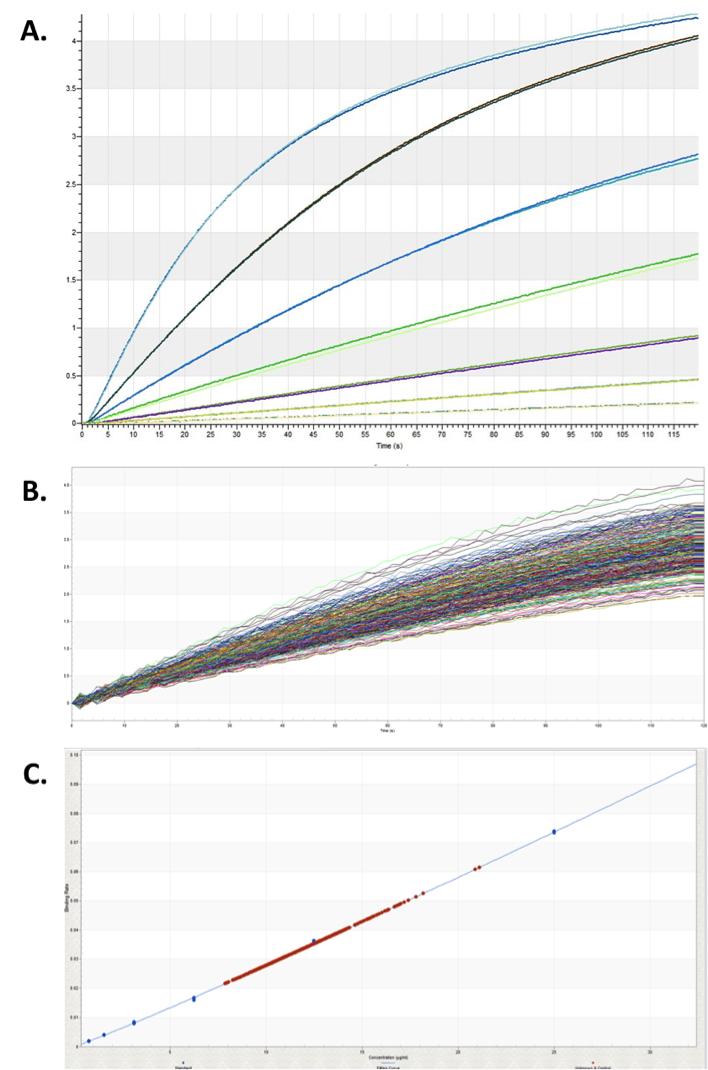
The 700 µg/mL Protein A Calibrator (Pall ForteBio) was serially diluted to a range of 0-50 µg/mL in OptiCHO and triplicate wells were assayed using Protein A biosensors. A standard curve was generated with these calibrators using the initial slope for the binding rate algorithm and a five parameter logistic curve (weighted Y2) to calculate the dose response from sample wells. Since sample wells would have different media makeups, each media additive was tested to with the calibrators to ensure they did not significantly affect the IgG signal or baseline.
Cell Growth Analysis
Upon initial plating and prior to sampling for IgG analysis, culture plates were imaged on a SpectraMax i3x Multi-Mode Platform with SpectraMax MiniMax 300 Imaging Cytometer (Figure 1). One brightfield image per well was acquired and cells were enumerated using StainFree Cell Detection Technology with the available CellsC setting.
Because changes in the morphology of CHO cell colonies under different media conditions could affect cell counts, we sought to verify cell growth results with an XTT assay. Following media sampling for IgG analysis the Biomek FXP Workstation added 15µL XTT reagent (Cell Signaling) to the cells and the culture plate was returned to 37°C. Absorbance was measured on the SpectraMax i3x Multi-Mode Platform at 475nm and 660nm after 1-4 hours of incubation.
Data Analysis
Replicate values were averaged and imported into Design-Expert 8.0.2 software (Stat-Ease) to identify factors that have significant effects on cell growth or IgG production, either alone or in combination with one another. In addition, Design-Expert was used to predict what conditions would maximize IgG production.
Results
The main goal of this optimization approach was to determine the conditions that will promote the highest level of IgG production. However, increased IgG production can be achieved by increasing the per-cell output and/or by accelerating cellular expansion. Hence we analyzed both cell growth and IgG titers to distinguish between these effects.
Image analysis of the plate 30 minutes after plating confirmed consistent cell numbers across the plate (CV = 6.1%), ensuring any differences detected are due to media composition. Figure 3 shows the 384 IgG titers following four days of incubation in 96 different media conditions plotted onto the IgG standard curve. One confounding factor in measuring IgG concentration was that evaporation reduced the volume in wells on the edge of the plate. The two highest outlier wells (Figure 3C) were both corner wells and quadruplicates with edge wells had a much higher CV (9.6%) than internal quadruplicates (2.9%). Because this effect was so significant, exterior wells were excluded from the calculations. Resulting average titers ranged from 7.8 to 14.6 µg/mL, with basal OptiCHO media producing 8.8 µg/mL IgG. This indicates that after a single round of media optimization we were able to identify conditions that could enhance IgG production by 66%.
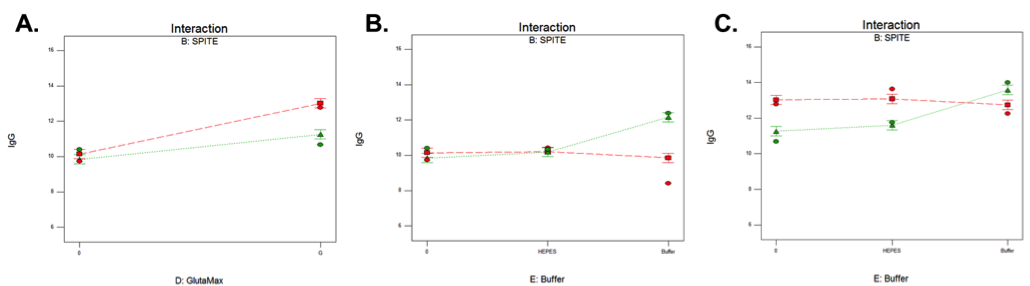
Statistical design software was used to identify the factors that had significant effects on IgG titer – either alone or in combination with one another (Table 1). In isolation, Polyamine and GlutaMAX increased IgG production, bioreactor buffer decreased production, and SPITE, HEPES, and EfficientFeed A had negligible effects. However, Figure 4 illustrates the significant interaction that SPITE had with both GlutaMAX and bioreactor buffer. SPITE actually reduced the positive effect of GlutaMAX in unbuffered conditions (Figure 4a) but SPITE combined with bioreactor buffer had significant positive effects (Figure 4b) and the highest IgG production was achieved by the combination of all three factors (Figure 4c). This shows the advantage of factorial analysis, as neither SPITE nor bioreactor buffer would have been taken forward if each factor was first assayed alone.
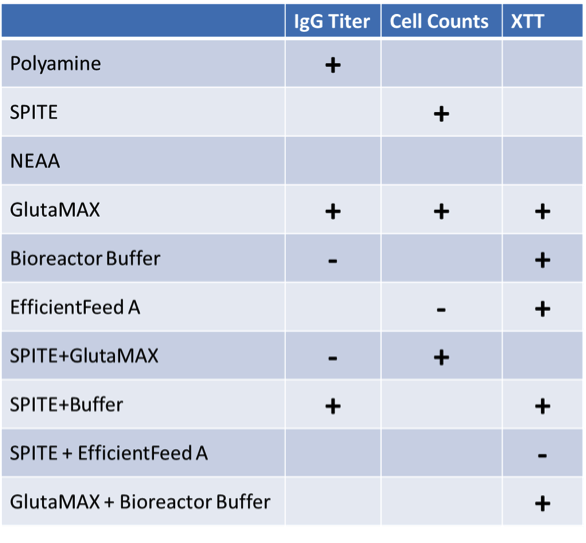
Increases indicated by “+” symbol, reductions indicated by “-“ symbol, and no symbol indicates no effect.
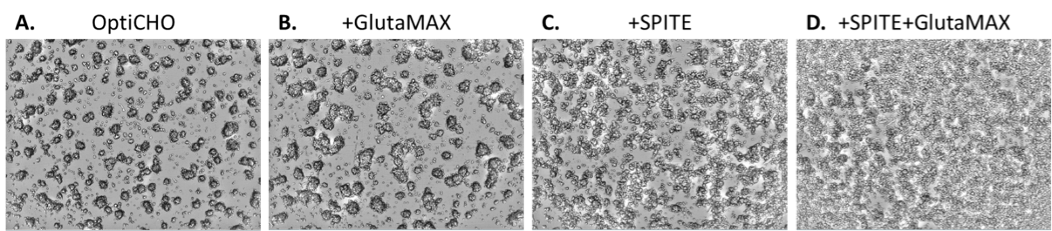
To investigate potential mechanisms of IgG increase, we analyzed effects of supplements on cell growth using the statistical design software. We found that GlutaMAX and SPITE significantly increased cell counts on the SpectraMax MiniMax cytometer individually and had a greater effect in combination while EfficientFeed A reduced cell counts (Table 1). Figure 5 shows that the CHO cells predominantly grew in three-dimensional clusters in OptiCHO media. GlutaMAX appears to increase the size of the colonies while SPITE seems to alter the colony morphology and/or increase the number of individual cells growing between colonies. Because of the three-dimensional nature of the cell colonies, changes in cell numbers could be under- or over-reported by imaging, therefore we utilized XTT assays as an additional means of quantifying the relative number of cells.
Again, to avoid the effects of evaporation on edge wells we only used interior wells for XTT analysis. Similar to the results we saw from cell imaging, GlutaMAX had a significant positive effect but SPITE only increased XTT absorbance in the presence of Bioreactor buffer. The effect of GlutaMAX was also increased in the presence of Bioreactor buffer. Interestingly, Efficient Feeder A was shown to have a positive effect on XTT absorbance but this effect was neutralized by the presence of SPITE. Finally, HEPES was shown to have a negative effect on XTT signal (Table 1). It is also possible that media differences could confound the XTT results (i.e. buffers/pH could affect the reaction chemistry) but for conditions such as GlutaMAX addition the reinforcement of imaging results provides stronger evidence of an effect on cell growth.
Finally, we used the statistical design software to create a model from the 96 data points to predict the optimal condition for IgG expression. This modeling reduces the likelihood of utilizing a “false positive” as the de facto best condition by simply taking the condition that gave the highest concentration in a single experiment. Based on the model, a media supplementation with Polyamine, SPITE, GlutaMAX, and Bioreactor pH Adjustment Solution would be predicted to generate 13.6 µg/mL IgG, a 42% increase over a predicted 9.6 µg/mL production in basal OptiCHO medium.
Discussion
Here we have shown how media optimization can improve CHO cell growth and protein production. By utilizing a factorial design of experiment we were able to identify conditions that improved antibody titer by 42-66% over a commercial medium that is optimized for CHO culture. We were also able to identify those factors that did not enhance expression and thereby reduce the costs of production cell culture by not including these supplements in the medium. By testing combinations of all supplements we were able to identify positive combinations that would not have been identified by testing supplements in single fashion. However, these factorial combinations are challenging to achieve in a manual fashion. This experiment would have required 184 manual transfers using a combination of single and 8-channel pipettes and this could grow significantly for more elaborate combinations. Not only does automation reduce the significant opportunity for errors in this approach, but the time a scientist would spend at the bench executing these steps can now be spent designing experiments and analyzing data.
By analyzing cell growth in addition to IgG titer, we were able to distinguish between factors that primarily affected cell growth (GlutaMAX, SPITE) from those that drove IgG production without affecting growth (Polyamine). This additional information suggests that a step-wise addition could be beneficial, whereby cell growth would first be stimulated, followed by a switch to protein production. In addition, there is likely a balance between these two effects, such that too much cell growth may reduce the energy available for producing IgG or so much energy is dedicated for protein production that cell growth slows. Once again, the factorial approach is more likely to identify the conditions that could provide the optimal balance.
Another consideration when utilizing this type of DOE approach is that the various supplements might affect the analysis. One example is that the addition of a Linoleic Acid/Oleic Acid/Albumin supplement (Sigma Aldrich) shifted the baseline on the Octet readings, even in the absence of cells or standards. Because not all wells would have this supplement, it would be difficult to identify legitimate increases in IgG production in these wells from false positives, and therefore this reagent was not included in the screen. As described above, it is also possible that different factors will have effects on the characteristics of the adherent cell colonies that could skew imaging cell counts or could affect the XTT reactions, thus it is useful to have multiple analyses when predicting mechanisms of action.
Other confounding factors include the evaporation effect in edge wells as described earlier. One way to minimize this effect is to look at shorter time periods. For example, at 2 days of culture, the replicate CVs for IgG titers for inner wells was 2.5% while those conditions with edge wells was only 3.8% (vs. 9.6% at day 4). However, longer culture times are more likely to identify conditions that might better translate to a bioreactor, where culture times can stretch to weeks. Options for minimizing edge effects include utilizing plates that have evaporation wells, or as we did here, eliminating the edge well values from consideration. One could also utilize the EST feature of the multichannel head of the Biomek FXP to only add reagents and cells to the 308 interior wells of the 384-well plate and then add water to the remaining 76 wells around the circumference of the plate. The EST function also allows the opportunity to leave wells available for the inclusion of a standard curve on each plate if that is preferable to utilizing a previously established standard curve for post-run analysis. A final approach to reducing evaporation effects would be to utilize the liquid level sensing capability of the Biomek Span-8 pipettors to calculate the volume of each well and then normalize the volumes prior to analysis, however, this would be a rate-limiting process compared to other steps in this IgG analysis.
Because of the ease of sample preparation and rapid processing of samples, the Octet HTX system is an excellent option for higher throughput applications such as expression clone screening. To enable a complete high-throughput workflow, we have integrated the Octet HTX system to the Biomek FXP (Figure 6). This configuration can automate the direct loading, processing, and analysis of protein quantification or kinetic studies. The system also has the ability to utilize the results to drive additional liquid handling steps, such as expanding high expressing clones to larger wells. By combining the liquid handling power of the Biomek FXP Workstation with the Octet HTX system one can minimize time, effort, and costs in protein production optimization and analysis.
Interview
After reading the paper, I was lucky to be able to interview two of the authors, Michael Kowalski and David Apiyo. Here is the summary of our interview:
Question:
This study focuses on optimizing media, what other applications would you use this equipment combination for? You touched on expression clone screening – could you expand?
Answer:
Michael explained that in looking for high expressing clones, researchers typically use either a cell sorting or limiting dilution approach. In either approach you could be processing thousands of wells trying to find the high expressing cells. Automation permits high throughput screening by automating the plating of cells, the maintenance of cells, and connecting to an integrated analyzer like the Octet system for analysis and clone selection.
David described that by using the Octet platform, researchers can identify the high producing clones, they can also identify optimal conditions. When screening thousands of clones, the Octet system allows clones to be divided into two main categories, high expressing clones that produce the highest concentration within a certain time period and best behaved clones for function and viability.
The Octet platform allows you to functionally qualify a specific clone by looking at antigen binding capability and specific affinity parameters.
Main areas of benefits for using the Biomek and the Octet systems together in cell line development and clone selection are
- High throughput screening of clones
- Viability, quality and functional analysis of the clones
- Epitope binning studies
- Formulation studies
- Advanced quantitation studies
Another area where these instruments could provide needed high throughput is in looking at host cell protein contamination. Being able to identify and quantifying host cell protein as you move from expression to purification would be extremely useful, but not possible without the high throughput capabilities that automation offers.
Question:
Could this technology be used for Cell Therapy media optimization?
Answer:
Michael explained that yes, the Biomek while for research only, couldn’t be used on the cells in Cell Therapy applications, it could however be used in upstream optimization. A similar DoE (design of experiment) approach as was used in this application note could be used to identify the best media conditions for a particular cell line.
Question:
How do you see companies implementing this technology in-house? What would the initial set up and training look like?
Answer:
Michael describes that the training requirements would vary based on the customer and customer need. In general, he likes to recommend that customers start simple and build to a more complex system as needed. Beckman Coulter and Pall ForteBio provide training and support, and a variety of different training methods are available, including on-site and off-site training for the Biomek.
The work that was done for this application note was done using a stand-alone Biomek and a stand-alone Octet HTX instrument. In this situation, by using automation, the authors were able to process the samples and then manually move the plate into the Octet system. This workflow was suitable for lower throughput demands. For higher throughput demand, like clone selection, the two machines can be integrated to provide a higher degree of automation. Additional benefits of integration would include reduced risk of human error, and in applications like David described, where the goal is to identify optimal cell lines and conditions, the Biomek could take the information from the Octet system and use it to drive additional pipetting to expand a colony or change other experimental conditions.
Question:
I think as important as finding out what worked, you also were able to identify what didn’t or had no impact on the cells, thereby allowing companies to eliminate ingredients and cut cost. Did you do any calculations of the potential time and cost savings of using your products for optimization?
Answer:
Michael described that most people take the media protocols and use them as is, even though there is a good chance that some of the components will have no effect on the selected cell line. Over time and over volume, the cost of these unnecessary components can add up. It behooves companies to really examine whether they need these components, and if so, it can also be determined accurately at what level they are needed.
David added that the time savings through automation is really a key benefit in this approach. Running these samples in another system could take more than a day, but using the Biomek and the Octet systems together, they were able to process them in 20 minutes. No one is going to disagree with the DoE approach for media development, but people don’t do it because it is time consuming and heavily manual. Automating the process makes this approach more appealing.