CELLvo™ Human Cord Blood-Endothelial Progenitor Cells are Highly Proliferative and Angiogenic
Introduction to Endothelial Progenitor Cells
Endothelial cells from the umbilical vein (hUVECs), artery (hAECs), and microvasculature (hMVECs) have been important in elucidating the pathways involved in angiogenesis and may have important clinical applications in the treatment of ischemic tissues or in the vascularization of graft tissues. In 2004, Mervin Yoder first described a novel population of rare circulating putative endothelial progenitor cells (EPCs) found in human umbilical cord blood and, less abundantly, in human adult peripheral blood [1]. These cells were highly proliferative relative to more commonly utilized endothelial-type cells. Yoder dubbed these putative endothelial progenitor cells as endothelial colony forming cells for their astounding capacity to form large primary, secondary, and tertiary colonies, as compared to other endothelial cell types. In addition to having superior colony forming ability, these cells have been shown to possess greater angiogenic potential in vitro as well as in vivo and it has been suggested that they may actually be progenitors of other endothelial cell types [2].
CELLvo™ Human Cord Blood-Endothelial Progenitor Cells
A major obstacle to the widespread adoption of endothelial progenitor cells for research use and therapeutic development has been their relative rarity, and difficulty isolating these cells from primary tissues. Isolation and expansion of this rare cell from human umbilical cord blood has been enabled by use of a proprietary and novel bone marrow-derived microenvironment, CELLvo™ Matrix. CELLvo™ Human Cord Blood – Endothelial Progenitor Cells (hCB-EPCs) are now commercially available, making their use in basic and translational research both practical and affordable.
With superior colony forming ability, greater vessel formation, and greater angiogenic potential as compared to more common endothelial-type cells (HUVECs) – CELLvo™ Human Cord Blood-Endothelial Progenitor Cells (hCB-EPCs) are the cell of choice for researchers. When grown on CELLvo™ Matrix, a bone marrow cell-derived extracellular matrix, CELLvo™ hCB-EPCs behave more naturally, allowing researchers greater control of their study.
CELLvo™ Human Cord Blood Endothelial Progenitor Cells compared to Human Umbilical Vein Endothelial Cells
StemBioSys CELLvo™ hCB-EPCs were directly compared to commercially available hUVECs. While they are similar in morphology and immunophenotype, CELLvo™ hCB-EPCs contrast to mature endothelial cells in many other ways. CELLvo™ hCB-EPCs form larger and more numerous primary colonies and go beyond primary colony formation, they have greater intrinsic vessel formation capacity, and demonstrate pro-angiogenic profiles relative to HUVECs as shown by in house studies. CELLvo™ hCB-EPCs previously isolated from fresh umbilical cord blood and expanded on CELLvo Matrix plates were directly compared to commercially available HUVECs. Cells were compared based on proliferation, phenotype, clonal expansion, vessel formation, and angiogenic paracrine profile. The results are described here in greater detail.
CELLvo™ hCB-EPCs form more colonies
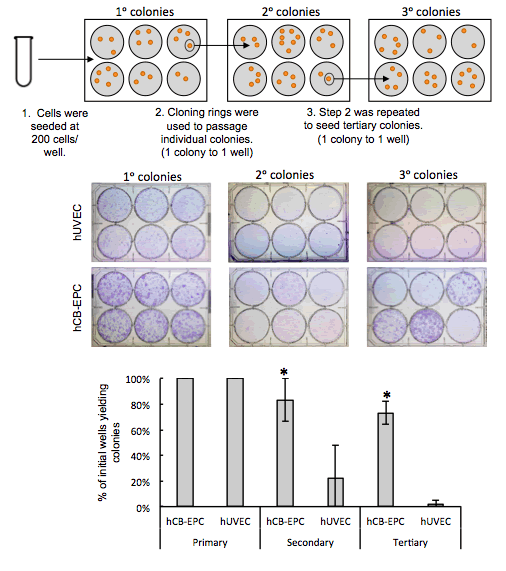
It has been reported that a key distinguishing characteristic of EPCs relative to more mature endothelial cell types such as HUVECs is their high proliferative potential especially as it relates to clonal expansion. CELLvo™ hCB-EPCs and hUVECs were seeded onto CELLvo™ Matrix at clonal density (20 cells/cm2). Individual colonies were detached and reseeded for secondary colony formation. The process was repeated for tertiary colony formation (Figure 1). It is important to note that both groups formed primary colonies consistently however over 80% of the CELLvo™ hCB-EPCs primary colonies were capable of secondary colony formation compared to the HUVECs with only 20%. Of those secondary colonies, nearly all of the CELLvo™ hCB-EPCs formed tertiary colonies. Of the secondary HUVECs, only 11% formed tertiary colonies.
CELLvo™ hCB-EPCs have greater intrinsic vessel formation
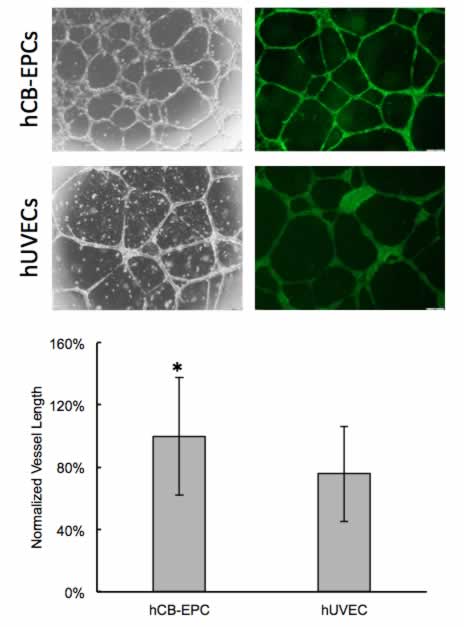
To measure intrinsic angiogenic properties of CELLvo™ hCB-EPCs, they were compared to hUVECs in a Matrigel® vessel formation assay. Following an 18 hour incubation, vessels spontaneously formed in the Matrigel® for both groups. Images of the vessel formation were analyzed using the NIH angiogenesis analyzer plug-in for ImageJ for non-biased quantification. CELLvo™ hCB-EPCs were significantly more angiogenic than hUVECs (as measured by total tube length per image) (Figure 2), forming approximately 25% more vessels (p=.025).
CELLvo™ hCB-EPCs demonstrate pro-angiogenic cell profiles
In addition to intrinsic vessel formation capacity, it may be therapeutically important for cells to secrete pro-angiogenic factors that may induce capillary formation via paracrine effects. This may be especially important, because mounting data suggests that tissue formation following cellular therapy is often carried out by endogenous cells, activated by growth factors from the transplanted cells. To test this, we used the RayBiotech Human Angiogenic Cytokine Array to examine the secretion of 43 pro-angiogenic cytokines into conditioned media of CELLvo™ hCB-EPCs and hUVECs. Of the 43 cytokines, 17 were expressed at higher levels by CELLvo™ hCB-EPCs relative to hUVECs. Only three were expressed at higher levels in conditioned media from hUVECs. There was no significant difference between the other 23 cytokines (Figure 3). Large differences were observed between the CELLvo™ hCB-EPCs and hUVECs in expression of VEGF, VEGF-R2, VEGF-R3, b-FGF, IL-10, GM-CSF, and I-309, among others.

Discussion
A great deal of work has been done with hUVECs and other mature endothelial cell types to understand angiogenesis and to develop potential therapies for the treatment of ischemic tissues. Recent work describing CELLvo™ hCB-EPCs as a novel population of cells that may be less mature than hUVECs, more pro-angiogenic, and have greater intrinsic vessel formation capacity is highly promising [3]. These cells may be more therapeutically useful in the treatment of ischemic tissues and may be useful in understanding how mature endothelial cell types are derived in vivo. It has even been suggested that a small fraction of these cells in more mature endothelial cell populations may be largely responsible for desirable functional attributes.
Though a growing excitement surrounds these cells, it has been difficult to isolate large quantities for research as they are relatively rare in cord blood, and extremely rare in adult peripheral blood. StemBioSys® methods make it possible to efficiently isolate hCB-EPCs from cord blood making them more available to researchers. This innovation makes it practical for research labs to shift away from hUVECs and hAECs to cells that are potentially much more potent. Here, we offer evidence supporting previous claims that StemBioSys® CELLvo™ hCB-EPCs are distinct from hUVECs and other commonly used endothelial cell types, and that they differ in terms of clonal expansion capability and angiogenic potential.
For more information, contact www.stembiosys.com
About the Author:

Travis Block, Ph.D.
Travis Block holds a Ph.D. in Biomedical Engineering from UT Health San Antonio. He has published papers on pharmacology, aging, Cell Therapy, and stem cell-matrix interactions. He holds patents related to adaptive equipment and cellular therapies. Currently, he serves as Senior Scientist at StemBioSys, Inc., a privately held biotech company in San Antonio, Texas. In this role, he works to develop novel tools for the stem cell and regenerative medicine field.
Acknowledgments: The laboratory support work, technical data analysis and images were provided by Mary Navarro MS, Anne Sheldrake MS, and Lucero Alvarado BS of StemBioSys, Inc.
Footnotes
-
1. Ingram, D. A. et al. Identification of a novel hierarchy of endothelial progenitor cells using human peripheral and umbilical cord blood. Blood 104, 2752–60 (2004).
-
2. Joo, H. et al. Human endothelial colony forming cells from adult peripheral blood have enhanced sprouting angiogenic potential through up-regulating VEGFR2 signaling. Int J Cardiol197, 33–43 (2015).
-
3. Critser, P. J. & Yoder, M. C. Endothelial colony-forming cell role in neoangiogenesis and tissue repair. Curr Opin Organ Transplant 15, 68–72 (2010).