Culturing hPSCs: From MEFs to Naïve State Cells
A Guest Blog by: Andrea Lai, PhD, Scientific Marketing Specialist and Simon Hilcove, PhD, Senior Product Marketing Manager, Pluripotent Stem Cells, STEMCELL Technologies
Contact: simon.hilcove@
Establishment of In Vitro hPSC Culture Conditions
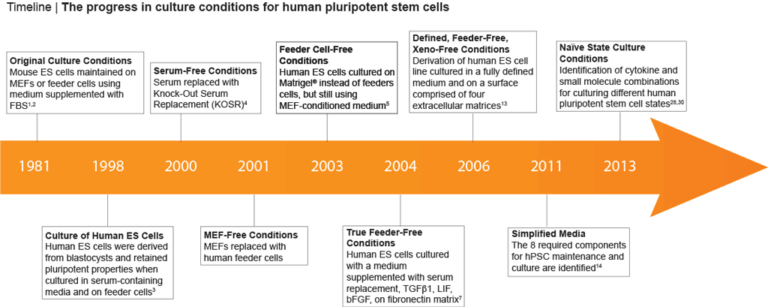
Culture conditions for human pluripotent stem cells (hPSCs) have improved greatly since in vitro methods for culturing human embryonic stem (hES) cells were first described (1,2) (Figure 1). These initial methods relied on the use of mouse embryonic fibroblasts (MEFs) or feeder cells in combination with fetal bovine serum (FBS) supplemented media. (3) However, the prospect of clinical uses for these cells quickly resulted in a need for improved methods that eliminate these potentially immunogenic animal-derived components. Efforts to improve hPSC culture protocols have largely focused on removing the undefined and non-human components (4-6) of the system (Figure 1), leading to feeder-free culture methods that utilize a combination of growth factors (bFGF and/or TGF-β), a serum replacement in the medium, and cultureware coated with an extracellular matrix surface such as Corning® Matrigel®. (7-8) Subsequently, multiple publications have reported formulations for the maintenance of hPSCs in a defined, feeder-free, or xeno-free (i.e. devoid of animal components) manner. (9-12)
Figure 1. Milestones in hPSC Culture
Defined, Feeder-Free Culture Conditions: Feeder Versus Feeder-Free
In 2006, Dr. Tenneille Ludwig of Dr. James Thomson’s lab, along with colleagues at the WiCell™ Research Institute (Madison, WI) reported the derivation of a new ES cell line in fully defined, feeder-free and xeno-free culture conditions. (12,13) Using a combination of bFGF to sustain self-renewal, TGF-β to inhibit differentiation and a number of other key factors to support pluripotency, this first defined medium significantly improved human ES cell culture. A modified version of this formulation, mTeSR™1, that includes all these is now the most widely-published feeder-free medium, and has been used in over 1100 peer-reviewed publications.
In 2011, Dr. Guokai Chen from Thomson lab developed a defined, simplified and low protein medium for the maintenance of hPSCs. (14) This formulation, known as E8, was established by identifying the 8 key components required for hPSC maintenance: insulin, selenium, transferrin, L-ascorbic acid, bFGF, TGFβ, NaHCO3, DMEM/F12. (14) For more information on the E8 Culture System for maintenance, cryopreservation and differentiation of hPSCs, watch this webinar with Dr. Guokai Chen.
Cell Attachment Substrates: Matrigel® Versus Vitronectin
mTeSR™1 was originally developed to work with hPSCs cultured on Matrigel®, a basement membrane matrix derived from Engelbreth-Holm-Swarm (EHS) mouse sarcoma cells. (12) However, Ludwig et al found that this was also a potential source of immunogenic proteins. A lot of effort has since focused on finding surface coatings that are more defined than Matrigel® but still support hPSC culture. Some of the more promising approaches are synthetic peptides chemically linked to the cultureware, (15,16) and recombinant proteins that interact with specific integrins and cell adhesion molecules. (17,18) Vitronectin XF™, a defined and xeno-free surface substrate, was developed and manufactured by Primorigen Biosciences and is an effective alternative to Matrigel® for hPSC maintenance. Compatible with all TeSR™ Family media, Vitronectin XF™ can be used with mTeSR™1 for fully defined culture, or with TeSR™2 or TeSR™-E8™ media for a completely xeno-free culture system.
Aggregate Versus Single-Cell Passaging
hPSCs have traditionally been passaged as small aggregates of approximately 50 – 200μm in diameter. These established methods have been shown to allow the expansion of many different cell lines for > 20 passages while maintaining a normal karyotype. Although it is possible to passage human ES and iPS cells as single cells, this is generally not recommended as it has been demonstrated that this practice can place unwanted selective pressure on cell populations and may lead to genetic aberrations in the culture. (19,20) If you prefer to passage cells in this manner, the karyotype of your culture should be checked frequently to ensure that your culture has retained a normal karyotype.
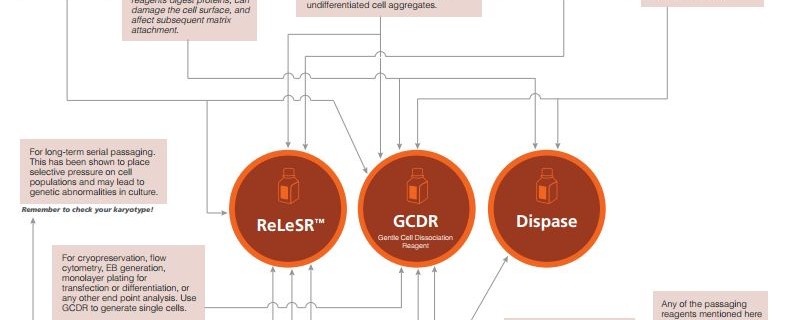
Making hPSC Passaging Easier: Enzyme Versus Enzyme-Free Passaging Reagents
hPSCs cultured under feeder-free conditions were initially passaged via mechanical scraping either with or without an enzymatic passaging reagent (e.g. Dispase) to break the colonies into small aggregates. However, this process is time-consuming and the breakup of aggregates to the desired size is subjective. Moreover, while enzymatic passaging reagents are effective, they digest extracellular proteins, can damage the cell surface, and need to be inactivated before replating.
Enzyme-free passaging reagents such as Gentle Cell Dissociation Reagent (GCDR) have since been developed to allow passaging while leaving cell surface proteins intact. Additionally the enzyme-inactivation step is not required which alleviates the need to centrifuge the cells during enzyme removal, a process that is time consuming and can lead to increased cell loss.
A recent advance that simplified the entire process of hPSC passaging was the development of another enzyme-free reagent, ReLeSR™. This passaging reagent improves consistency and reproducibility of hPSC culture by allowing for the selective detachment of undifferentiated cells in optimally-sized aggregates, negating the need for manual scraping, enzymatic detachment, and break-up of large aggregates. This allows for significant time savings, especially if ReLeSR™ is used for passaging large numbers of cells in flasks or other closed vessels. Examples of the different applications where ReLeSR™ can be used (such as isolation of newly reprogrammed iPS cell colonies) can be found in our Innovation Showcase talk from the ISSCR 2014 Annual Meeting.
Large-Scale hPSC Culture: 2D Versus 3D Cell Culture
The use of hPSCs for applications such as drug screening, toxicological testing or cell-based therapies medicine requires cultivation of large numbers of cells. Until recently, the manual steps involved in hPSC passaging were one obstacle to easy scale-up of 2D hPSC culture. However, while ReLeSR™ will facilitate culture scale-up to larger flasks or cell factories, the cell numbers required for certain clinical applications still exceed what can be generated in 2D culture. In a 3D environment, hPSCs can be cultured on microcarriers or as aggregates, in spinner flasks or large stirred tank bioreactors. (21-25) The requirements for hPSC suspension culture are not the same as those for adherent cultures. For example, hPSCs can be cultured as aggregates without matrix attachment (a requirement in 2D culture) and feeding strategies (i.e. perfusion- versus batch-feeding) are of greater importance in a large-scale bioreactor. As such, many groups are still optimizing protocols for large-scale hPSC production to improve yield, robustness and efficiency.
Culture Requirements for Naïve State hPSCs: Naïve-like Versus Primed
Human PSCs differ at the developmental level from mouse ES cells and are considered to more closely resemble post-implantation mouse epiblast stem cells (EpiSCs) (26) or “primed state” PSCs (Table 1). In comparison, mouse ES cells are acknowledged to be in a more naive or ground state of pluripotency. Recently, multiple groups have demonstrated that mouse ES cells and conventional hPSCs exhibit distinct gene expression patterns and have different requirements in culture. (27-29) Specifically, maintenance of mouse ES cells in the undifferentiated state is dependent on culture conditions that contain leukemia inhibitory factor (LIF) whereas hPSCs or mouse EpiSCs are cultured in FGF and Activin and are not responsive to LIF. (27) Several research groups have recently identified culture conditions capable of reverting and maintaining human PSCs in a “ground state” as opposed to the “primed state” that hPSCs are traditionally maintained in. (28, 30-32) While we are learning more about the spectrum of pluripotency states and the processes that allow us to shift cells between these states, there are still many unknowns in this exciting and rapidly expanding area of hPSC research.
Table 1. Comparison of Naive-Like and Primed Pluripotent Stem Cells
To learn more about naive-like hPSCs, watch Dr. Huck-Hui Ng’s talk from the ISSCR 2014 Annual Meeting about using small molecules to shift primed hPSCs to a more native state, or request a free wallchart from Nature Reviews Molecular Cell Biology on naive and primed stem cell states.
More Options Than Ever
It is clear that many advances have been made over the past few decades to improve, define and standardize hPSC culture. These improvements have increased consistency, simplified hPSC maintenance, facilitated culture scale-up and helped to standardize procedures as we move closer to utilizing stem cells in therapeutics, drug screening and toxicity testing. However, these advances have also led to the arrival of more options than ever for maintenance media, matrices, passaging reagents, and cryopreservation media. Choosing the right set of conditions for your needs and applications can be daunting. Use our infographics to help decide which set of reagents will suit your hPSC culture needs best.
References
- Evans MJ & Kaufman MH. (1981) Establishment in culture of pluripotential cells from mouse embryos. Nature 292(5819): 154–6.
- Martin GR. (1981) Isolation of a pluripotent cell line from early mouse embryos cultured in medium conditioned by teratocarcinoma stem cells. Proc Natl Acad Sci U S A 78(12): 7634–8.
- Thomson JA et al. (1998) Embryonic stem cell lines derived from human blastocysts. Science 282(5391): 1145–7.
- Amit M et al. (2000) Clonally derived human embryonic stem cell lines maintain pluripotency and proliferative potential for prolonged periods of culture. Dev Biol 227(2): 271–8.
- Hovatta O et al. (2003) A culture system using human foreskin fibroblasts as feeder cells allows production of human embryonic stem cells. Hum Reprod 18(7): 1404–9.
- Xu C et al. (2001) Feeder-free growth of undifferentiated human embryonic stem cells. Nat Biotechnol 19(10): 971–4.
- Amit M et al. (2004) Feeder layer- and serum-free culture of human embryonic stem cells. Biol Reprod 70(3): 837–845.
- Levenstein ME et al. (2006) Basic fibroblast growth factor support of human embryonic stem cell self-renewal. Stem Cells 24(3): 568–74.
- Vallier L et al. (2005) Activin/Nodal and FGF pathways cooperate to maintain pluripotency of human embryonic stem cells. J Cell Sci 118(19): 4495–509.
- Lu J et al. (2006) Defined culture conditions of human embryonic stem cells. Proc Natl Acad Sci U S A 103(15): 5688–93.
- Yao S et al. (2006) Long-term self-renewal and directed differentiation of human embryonic stem cells in chemically defined conditions. Proc Natl Acad Sci U S A 103(18): 6907–12.
- Ludwig TE et al. (2006) Feeder-independent culture of human embryonic stem cells. Nat Methods 3(8): 637–46.
- Ludwig TE et al. (2006) Derivation of human embryonic stem cells in defined conditions. Nat Biotechnol 24(2): 185–7.
- Chen G et al. (2011) Chemically defined conditions for human iPSC derivation and culture. Nat Methods 8(5): 424–9.
- Melkoumian Z et al. (2010) Synthetic peptide-acrylate surfaces for long-term self-renewal and cardiomyocyte differentiation of human embryonic stem cells. Nat Biotechnol 28(6): 606–10.
- Villa-Diaz LG et al. (2010) Synthetic polymer coatings for long-term growth of human embryonic stem cells. Nat Biotechnol 28(6): 581–3.
- Rodin S et al. (2010) Long-term self-renewal of human pluripotent stem cells on human recombinant laminin-511. Nat Biotechnol 28(6): 611–5.
- Nagaoka M et al. (2006) E-cadherin-coated plates maintain pluripotent ES cells without colony formation. PLoS One 1: e15.
- Draper JS et al. (2004) Recurrent gain of chromosomes 17q and 12 in cultured human embryonic stem cells. Nat Biotechnol 22(1): 53–4.
- Buzzard JJ et al. (2004) Karyotype of human ES cells during extended culture. Nat Biotechnol 22(4): 381–2; author reply 382.
- Olmer R et al. (2010) Long term expansion of undifferentiated human iPS and ES cells in suspension culture using a defined medium. Stem Cell Res 5(1): 51–64.
- Oh SKW et al. (2009) Long-term microcarrier suspension cultures of human embryonic stem cells. Stem Cell Res 2(3): 219–230.
- Singh H et al. (2010) Up-scaling single cell-inoculated suspension culture of human embryonic stem cells. Stem Cell Res 4(3): 165–79.
- Krawetz R et al. (2010) Large-Scale Expansion of Pluripotent Human Embryonic Stem Cells in Stirred-Suspension Bioreactors.
- Zweigerdt R et al. (2011) Scalable expansion of human pluripotent stem cells in suspension culture. Nat Protoc 6(5): 689–700.
- Rossant J. (2008) Stem Cells and Early Lineage Development. Cell 132(4): 527–531.
- Nichols J et al. (1998) Formation of pluripotent stem cells in the mammalian embryo depends on the POU transcription factor Oct4. Cell 95(3): 379–391.
- Takashima Y et al. (2014) Resetting Transcription Factor Control Circuitry toward Ground-State Pluripotency in Human. Cell 158(6): 1254–1269.
- Marks H et al. (2012) The transcriptional and epigenomic foundations of ground state pluripotency. Cell 149(3): 590–604.
- Gafni O et al. (2013) Derivation of novel human ground state naive pluripotent stem cells. Nature 504(7479): 282–6.
- Theunissen TW et al. (2014) Systematic Identification of Defined Conditions for Induction and Maintenance of Naive Human Pluripotency. Cell Stem Cell 15(4): 471–487.
- Chan YS et al. (2013) Induction of a human pluripotent state with distinct regulatory circuitry that resembles preimplantation epiblast. Cell Stem Cell 13(6): 663–675.