
Enhancing Biopharmaceutical Production: The Impact of Chemically Defined Peptides on Biopharma Upstream Process Efficiency and Cost
Cell culture has become an essential part of biopharmaceutical manufacturing for the production of recombinant proteins, antibodies, vaccines, and gene therapies for a range of therapeutic indications. As a result, cell culture media formulations have evolved significantly to meet the needs of the industry. In particular, today’s formulations must be chemically defined and cannot contain animal-derived components. This can be achieved by replacing complex components with amino acids, vitamins, and other ingredients. The shift towards non-animal-derived formulations is driven by the overall need to increase product safety, reproducibility, and regulatory compliance. In addition, as production cell lines become more specialized, there is a growing need for customizable media formulations and higher nutrient concentrations to increase productivity by intensifying and optimizing biopharmaceutical processes, especially in monoclonal antibody production platforms.
Amino acids are an essential component of cell culture media, serving as the primary building blocks for protein synthesis. However, some commonly used amino acids pose challenges to the current media formulation landscape. Tyrosine, cystine and glutamine are examples of amino acids that either have low solubility, are chemically reactive, or are generally unstable, which can limit their bioavailability to the cells of interest. These properties also limit process performance, especially if highly concentrated feed or perfusion media are required to intensify the process. To overcome the challenges posed by the physicochemical limitations of amino acids, chemically defined peptides have emerged as an ideal choice for industrial cell culture media. These peptides offer several advantages, including the elimination of animal-derived components as well as improved stability and solubility compared to free amino acids.
Small differences in raw material quality can have a significant impact on bioprocess productivity. That’s why working with a trusted and reputable manufacturer is essential to mitigate risks and support overall success in biopharmaceutical production. Evonik is a leading global provider of high-quality cell culture ingredients, including peptides, amino and keto acids, lipids, and carbohydrates, with a robust pharmaceutical-grade quality system and extensive expertise in the manufacture of commercial-scale, chemically defined material that meets the highest purity and regulatory standards.
Evonik cQrex® Portfolio
Evonik has developed the cQrex® portfolio of chemically defined, highly pure and cGMP peptides and keto acids that can be used to improve the reproducibility and efficiency of bioprocesses. Peptides are molecules formed by the chemical coupling of amino acids to improve the stability, solubility, and bioavailability of target amino acids. A peptide bond is formed by a condensation reaction between the carboxyl group (-COOH) of one amino acid and the amino group (-NH2) of another amino acid, resulting in the release of a water molecule.
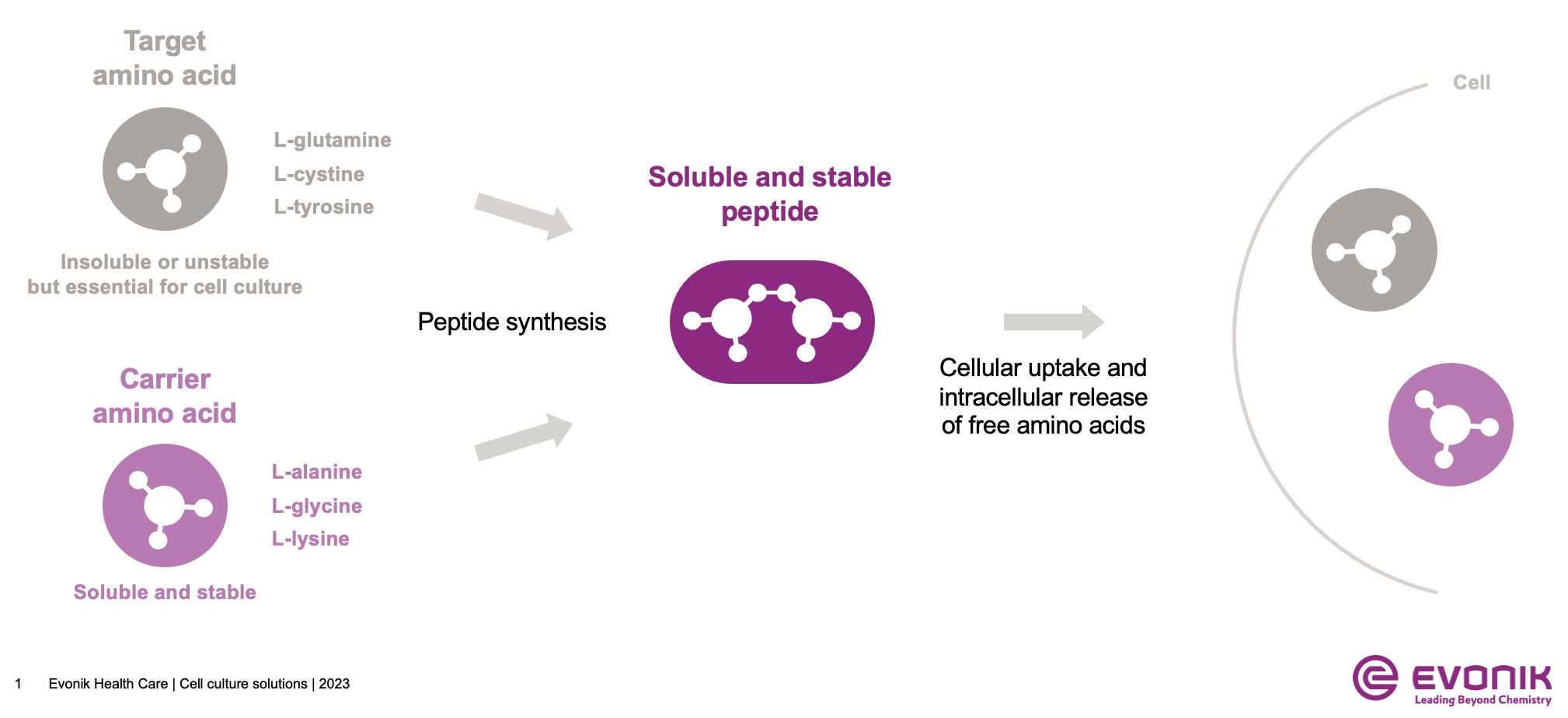
1) L-Glutamine Stability
L-glutamine is an essential amino acid and is required in cell culture media as a carbon source for growth. Due to its important role in energy supply, free L-glutamine is present in cell culture media at higher concentrations than other amino acids. However, its limited chemical stability in aqueous solutions makes it susceptible to degradation, leading to the formation of byproducts like ammonia, glutamic acid, or pyroglutamate, which can negatively affect cell culture performance.
Using freshly prepared solutions of L-glutamine is a common strategy to compensate for its chemical stability issues, but this complicates the process with additional steps. Given these challenges, the use of stabilized dipeptide forms of L-glutamine such as cQrex® GQ (Glycyl-L-Glutamine hydrate) and cQrex® AQ (L-Alanyl-L-Glutamine) offers a more stable and controlled alternative. cQrex® GQ and cQrex® AQ are stable in stock solutions and media for at least two months at ambient temperature, thus reducing processing and usage complexities.
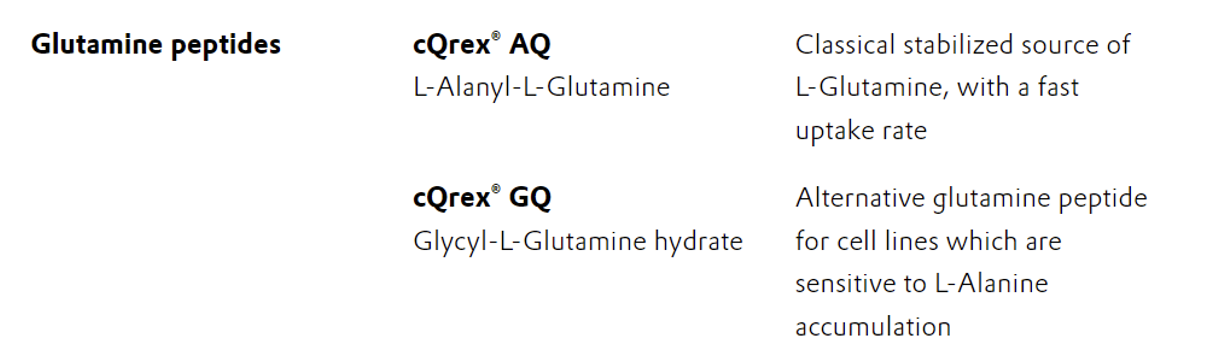
2) L-Tyrosine and L-Cystine Solubility
L-Tyrosine and L-cystine are known to be poorly soluble in water and aqueous media. The use of extreme pH values is often required to overcome solubility limitations and achieve the desired concentration of these amino acids. For example, separate alkaline feeds containing L-tyrosine and L-cystine are traditionally used in addition to the main pH-neutral feed to improve cell performance and batch productivity. However, this mitigation strategy has many drawbacks that can adversely affect cell viability and protein production. Not only does the separate feed increase process complexity, but it can also introduce additional risks with high salt concentrations, potential pH spikes, and the precipitation of L-tyrosine and L-cystine in the bioreactor.
Substitution of L-tyrosine and L-cystine with peptides can effectively increase the solubility at neutral pH by up to 1000-fold, and because these peptides can be efficiently taken up and metabolized by the cell, full replacement of the amino acids in media formulations is also possible.
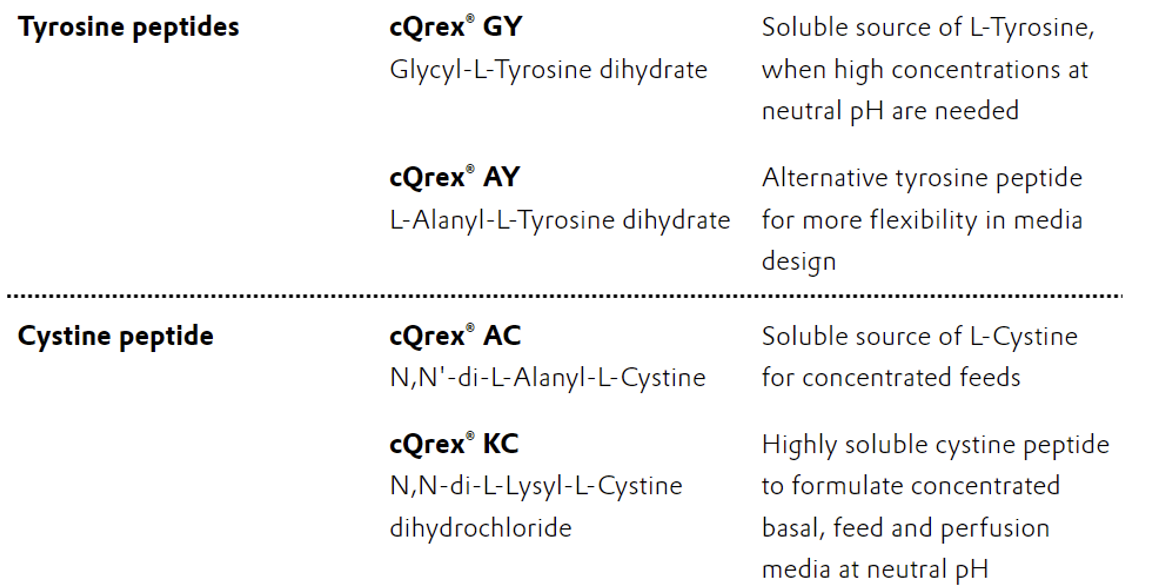
cQrex® KC (N,N‘-di-L-Lysyl-L-Cystine dihydrochloride) is a recent addition to the peptide portfolio that is 1000 times more soluble than L-cystine. Serving as a precursor for crucial amino acids like L-cystine and L-lysine, cQrex® KC enables the formulation of highly concentrated and pH-neutral cell culture media. By increasing the cell viability and cell-specific productivity, cQrex® KC also contributes to higher antibody titers and increased overall productivity of biological drugs.
3) Optimization of the upstream processing and production cost reduction
Replacement of amino acids with highly stable and soluble cQrex® peptides can solve common cell culture media and bioprocessing challenges to enable:
- Process intensification: Formulate highly concentrated and stable basal, feed and perfusion media.
- Process simplification: Leverage stable and highly soluble peptides at neutral pH to remove alkaline feeds from the bioprocess.
- Increased productivity: Tailor specific nutrient requirements and use performance-boosting ingredients for highly efficient biopharmaceuticals production.
In addition to the clear advantages of stability and solubility, the cQrex® peptides help improve media formulations to overcome bioprocess limitations. If the focus is on increasing the concentration in the feed or perfusion media, the high solubility of the peptides allows the formulation of media with high concentrations of amino acid forms. Where media performance is unsatisfactory, the addition of peptides as supplements or the reformulation of media with peptides can lead to improved performance. For example, studies have shown that cQrex® KC boosts cell metabolism and enables a higher antibody titer.
Moreover, users can look for solutions to avoid complex two-feed systems. Thanks to their high solubility at neutral pH, cQrex® peptides enable the formulation of one-feed systems. Various options are possible and Evonik experts can advise on which to choose. All options result in a significant simplification of the upstream biopharma processing and reduced risks.
In summary, by adding the cQrex® peptides to the basal, feed and perfusion media, higher nutrient concentration and bioprocess simplification can be achieved. This leads to an increased productivity of biopharmaceuticals, a more sustainable process, and a reduction in overall production costs.

Evonik Services
Evonik’s global team of technical experts can provide comprehensive support, including application guidelines, product documentation, and a range of regulatory and quality services, to facilitate seamless implementation.
Evonik is committed to serving as a reliable supply partner by providing high-quality cell culture ingredients and value-adding services to mitigate risk, improve reproducibility, increase productivity, and strengthen supply security.
For more information on cQrex® ingredients, please visit: https://healthcare.evonik.com/en/biopharma/cell-culture/cqrex-portfolio
To request an initial consultation or request free samples to conduct initial feasibility studies, contact Evonik at healthcare@evonik.com