Generation, Maintenance, and Cryopreservation of Neural Progenitor Cells derived from Human Pluripotent Stem Cells using the STEMdiff Neural System
Vivian M. Lee, Alexandra Blak, Melanie A. Olson, Jennifer Antonchuck, Allen C. Eaves, Terry E. Thomas and Sharon A. Louis
STEMCELL Technologies
Introduction:
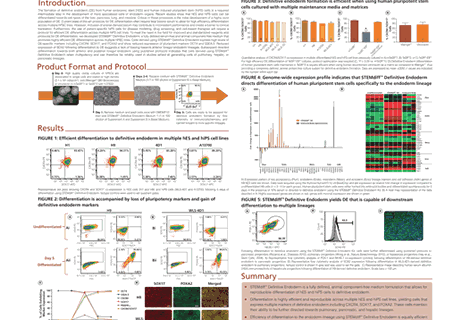
Neural progenitor cells (NPCs) generated from human pluripotent stem cells (hPSC), including human embryonic stem (hES) cells and induced pluripotent stem (hiPS) cells are extensively used for studying human nervous system development, modeling neurological disorders, and screening for therapeutic molecules. We have reported that CNS-specific NPCs can be produced efficiently using STEMdiff™ Neural Induction Medium (NIM) in conjunction with AggreWell™800. Recent publications have shown that neural induction from hPSCs can also be achieved in a single-step monolayer culture system, which does not require aggregate formation and therefore simplifies the process of neural induction. Here we describe: 1) the use of STEMdiff™ NIM in a monolayer-based neural induction system and 2) the use of two new defined media for propagating [STEMdiff™ Neural Progenitor Medium (NPM)] and cryopreserving [STEMdiff™ Neural Progenitor Freezing Medium (NPFM)] NPCs, respectively. Toward the first objective, hPSCs maintained in mTeSR™1 were seeded at 250,000 cells/cm2 and cultured as monolayers in NIM for up to 10 days. Cells were assessed at different time points for neural induction by the appearance of PAX6 with concomitant down regulation of OCT4. In the hES cell lines tested, approximately 25 – 30% of the cells were PAX6+0CT4– by day 3 and all cells were PAX6+0CT 4–by day 6 (n = 5). In the hiPS cell lines tested, 30 – 40% of cells were PAX6+0CT 4– on day 5 and induction to PAX6+0CT 4– cells was complete by day 9 (n = 4). These data show that CNS-specific NPCs can be efficiently produced in a single-step from hPSC using STEMdiff™ NIM with a monolayer protocol.
For the second objective, we generated NPCs using STEMdiff™ NIM in either the standard AggreWell™800 system or the monolayer system. NPCs were then dissociated into single-cell suspensions and either cultured further in NPM or cryopreserved in NPFM. Cells were passaged in STEMdiff™ NPM every 7 days and cell expansion was calculated at each passage. Overall NPCs propagated in NPM exhibited 3 – 5 fold expansion (n = 8) over the first 3 passages. NPCs were maintained in NPM with low spontaneous neuronal differentiation <10%) for at least 10 passages. Lastly, we showed that NPCs generated using NIM could be cryopreserved in NPFM, with high viability and recovery rate upon thawing. Thawed NPCs could be further expanded or used for differentiation experiments.
We have now expanded the application of STEMdiff™ NIM in a monolayer-culture based protocol for neural induction. With this, our STEMdiff™ Neural is an integrated system for the generation, maintenance, and cryopreservation of hPSC-derived NPCs.