Intestinal Organoid Culture: a Convenient and Physiologically Relevant Model for Intestinal and Stem Cell Research
Guest blog by: Ryan Conder, PhD, Senior Scientist, Epithelial Cell Biology, STEMCELL Technologies
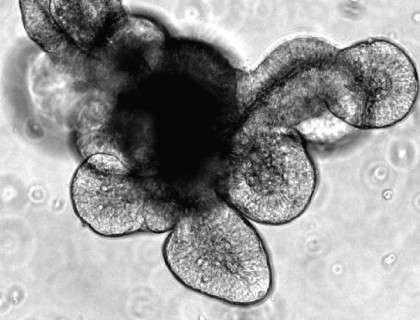
Intestinal epithelial organoids are functional three dimensional (3D) models of the intestine that are novel and exciting tools for research across multiple disciplines including intestinal biology, adult stem cell biology and immunology. The 3D multicellular structures of intestinal organoids retain key physiological features of an intestine: the crypt and villus domains, a functional lumen and all of the major cell types present in the adult intestinal epithelium. The organoid culture can be passaged, expanded and frozen, and can be genetically manipulated via transfection of DNA or siRNA, or infection with an adeno- or lentivirus. As the organoids contain intestinal stem cells that rapidly divide and differentiate, this culture system is a convenient model for researching regenerative therapies. By bridging the gap between currently applied in vivo and in vitro methods, intestinal organoids are allowing scientists to ask and investigate questions that were not previously possible.
IntestiCult™ Organoid Growth Medium (Mouse) is the first-ever complete, defined, serum-free medium for generating intestinal organoids from mouse intestinal crypts.
The Intestinal Epithelium
The mammalian intestine is lined with a single layer of epithelial cells that are assigned to one of two domains: a basal crypt domain responsible for the maintenance and regeneration of the epithelium, and the extruding villus domain that is primarily involved in the absorption of nutrients from the intestinal lumen.(1,2) The crypt domain consists of three main cell types: intestinal stem cells (ISCs), which express LGR5 (a protein in the WNT signaling pathway) and differentiate to give rise to all other intestinal epithelial cell types; Paneth cells, which secrete cytokines to maintain the stem cell niche and secrete antimicrobial peptides as a part of the innate immune response; and transit amplifying cells, which are partially differentiated cells that terminally differentiate into the cell types found in the villus. There are also three main cell types found in the villus domain: goblet cells, which secrete mucus to protect the epithelium and to help move intestinal contents through the lumen; enteroendocrine cells, which secrete hormones to regulate nutrient metabolism; and enterocytes, which form the majority of the villus and are responsible for nutrient uptake.
In mammals, the intestinal epithelium undergoes complete self-renewal every four to five days thanks to the rapid division and differentiation of ISCs.(3) Depending on their location within the crypt, ISCs either renew the stem cell population or begin differentiating by becoming a part of the rapidly dividing transit amplifying population. ISCs divide symmetrically within a spatially restricted niche, leading to the removal of some ISCs from the niche while other ISCs retain their position at the crypt base and maintain their “stemness”.(4) Over time, the ISCs in a given crypt drift toward clonality as the progeny of neighboring stem cells are excluded from the niche. While recent lineage-tracing experiments have identified ISCs as being located at the crypt base, further studies have demonstrated that ISC populations are present outside of the crypt, specifically at the +4 position, and can revert to multipotent ISCs upon intestinal damage.(5) These findings demonstrate the plasticity of the stem and progenitor cells in the intestinal tissue that is necessary for maintaining homeostasis in the harsh environment of the intestinal lumen. Furthermore, these regenerative properties make the intestinal epithelium an attractive model system to study general characteristics of both epithelial tissues and adult stem cells, as well as mechanisms specific to the intestinal epithelium and intestinal diseases.
Current Challenges in Intestinal Research
Studying the intestinal epithelium can pose multiple challenges. In vitro models of the intestine are convenient for investigating the impact of specific variables on intestinal cells, but lack many key structural features of the in vivo epithelium. One such approach is using cell lines derived from intestinal tumors, such as the CACO-2 adenocarcinoma line, which is commonly used due to its ability to spontaneously differentiate and produce a monolayer of enterocytes.(6) However, such cells lack the genetic and cellular diversity found in the in vivo intestine, and the resulting culture closely resembles cells of the fetal, rather than adult, intestine. Immortalization of these cell lines can also lead to functionally significant genetic differences between the cell lines being assayed and the source tissue. These approaches involve the use of monolayer two-dimensional cultures which limit the physiological relevance for certain experiments.
In vivo models have the advantage of direct experimentation on the intact intestine. More specifically, using a mouse model is beneficial due to the similarities between the human and mouse intestine, such as the conserved LGR5 marker protein found in the ISCs of both species.(7) However, studying the intestine in the whole animal also has its disadvantages. It is time consuming and expensive to run in vivo experiments and they are not easily replicated in a human system. It is also difficult to isolate the effects specifically pertaining to the intestinal epithelium when using an intact mouse as external variables can be introduced from the mesenchymal tissue or the immune, circulatory, or endocrine systems.
In comparison, intestinal epithelial organoids are 3D cultures containing the isolated epithelium with a crypt-villus structure, functional lumen and all of the major cell types found in the adult intestinal epithelium. Due to its convenience and high physiological relevance, intestinal organoid culture is a powerful research tool that enables researchers to address many of the current challenges in intestinal research.
History of Intestinal Organoids
Early attempts to produce an ex vivo culture system of the intestinal epithelium resulted in models where the ISCs could transiently divide but would lose the ability to proliferate within weeks.(8,9) In 2009, Dr. Calvin Kuo and his group published the first successful culture system that could promote long-term survival of ISCs: cyst-like structures containing all major intestinal cell types derived from neonatal mouse intestinal fragments containing epithelial and mesenchymal cells.(10) While the cultures could be maintained for over one year, the cultures lacked the crypt-villus features of the intestine, and the process of generating these structures involved devising an air-liquid interface and using fetal bovine serum.(10)
An alternative technique was published the same year by Dr. Hans Clevers’ group in which crypt-villus “organoid” structures were successfully generated from both isolated mouse intestinal crypts and single sorted LGR5+ ISCs.(11) In lieu of the air-liquid interface, the system used a semi-solid, laminin/collagen-rich Matrigel® dome and a specialized cell culture medium to mimic the extracellular matrix and stem cell niche, respectively, and enable long-term culture of the cells. The organoid structure contained a central lumen surrounded by “buds” representing intestinal crypts which were functionally similar to the adult intestinal crypt, as dividing LGR5+ ISCs were present and intercalated between Paneth cells. The ISCs were also able to differentiate into goblet cells, enteroendocrine cells, or enterocytes and these terminally differentiated cells would ultimately extrude into the lumen to form the villus-like domain. Overall, the organoid culture produced by the Clevers group had a high degree of physiological relevance, and could also be maintained indefinitely with periodic cell dissociation and passaging.(11) This advancement represented the establishment of a novel model system that incorporates many physiological properties of in vivo mouse models with easy-to-use methodologies that are typical of in vitro cell culture models.
Research Applications of Intestinal Organoids
Although intestinal organoid technology is still in its infancy, this new culture system has already been demonstrated as a valuable research model and diverse proof-of-concept investigations validate its wide variety of applications. Organoids grown from mouse intestinal crypts can be subjected to changes in gene expression through transfection or viral infection.(12) The rapid expansion of organoids in culture allows for their use in various analytical procedures including microarrays, sequencing, immunohistochemistry and mass spectrometry.(13,14) Organoids can also be cryopreserved for long-term storage. Below are some recent examples of how intestinal organoid culture is being applied:
Intestinal Stem Cell Biology
Intestinal organoids have been used to identify a complex role for Paneth cells in stem cell maintenance as depletion of Paneth cells corresponds with a loss of LGR5+ cells from the crypt base.(15) These cells have also been demonstrated to exhibit differential sensitivity to bacterial ligands, cytokines and muscarinergic stimulation.(15,16) As a clearer understanding of the specific roles of the individual cell types is established, it may be possible to target cells for treatment of diseases arising from their abnormalities, such as targeting Paneth cell mutations in inflammatory bowel disease.(16)
Intestinal Disease
Intestinal disorders such as Crohn’s disease, ulcerative colitis, coeliac disease and sepsis result in uncontrolled activation of the immune system, leading to intestinal damage. Microscopy and colorimetric assays using intestinal organoids have been designed to study the mechanisms involved in the cell death pathways of these epithelial cells.(17) The unique architecture of organoids (i.e. an isolated epithelium and a functional lumen) also allows antimicrobial responses specific to Paneth cells to be studied. One such study involved establishing an enteric infection model where a bacterial pathogen was directly injected into the lumen of the organoids.(18) This technical development offers a novel model to investigate host-microbe interactions, whether with pathogenic or commensal microorganisms.
Adult Stem Cell Biology
Applications of intestinal organoids are not limited to intestinal research. The continual renewal of the intestinal tissue from the division and differentiation of ISCs makes this system ideal for studying the molecular mechanisms and pathways that are characteristic of adult stem cells at large. Research that is routinely carried out using Drosophila melanogaster or Caenorhabditis elegans as model organisms can be further explored using mouse intestinal organoid cultures to validate the functional homology of molecular pathways and interactions in an easily accessible mammalian system.
Gene Therapy
Organoids can be subjected to viral infection with specific vectors, and can also be transplanted into recipient mice after intestinal damage has been induced.(19) These two attributes make it possible to address questions involving the correction of specific disease-causing mutations. Because organoids can be grown from single stem cells, clonal organoids can be generated and analyzed after transfection. This allows for precise transplantation of organoids that have been identified as exhibiting proper integration of introduced genetic material and makes a strong case for organoids as a cell-based therapeutic tool. Two recent studies have focused on assay development and subsequent gene replacement therapy of the Cystic Fibrosis Transmembrane Conductance Receptor (CFTR) using organoid cultures as integral components.(20,21)
Drug-Screening
Beyond studies looking at specific signaling pathways, organoids also offer an opportunity for large-scale drug screening. Organoid-based screening can be either complementary to two-dimensional screens performed using CACO-2 cells, or serve to replace these cell line-based assays with a more physiologically relevant model system.(22) Proof-of-principle experiments using organoids to investigate the effect of small molecules on stem cell physiology have been performed.
Personalized Treatment
The ability to grow organoids that are genetically identical to individual patients represents a monumental step toward the elusive promise of individual patient disease diagnoses. Organoid cultures can be generated from normal and tumorous human intestines, a feature that makes it conceptually possible to use these cultures to screen for specific tumor-associated mutations and formulate more personalized treatment options. The HUB Foundation for Organoid Technology (http://hub4organoids.eu/) is in the process of generating a human organoid library, or “biobank”, composed of matched healthy and diseased patient biopsies that can be subjected to multiple screening methods.
Incorporating Intestinal Organoids into Your Research
Development of a cell culture medium that allows for the crypt-like structures of intestinal organoids to maintain their stemness has been a key factor in enabling successful culture of organoids. The intestinal stem cell niche requires a precise gradient of activity from multiple signaling pathways to maintain the proper homeostasis of the intestine. Intestinal organoids are generated from freshly isolated intestinal crypts that are cultured in a semi-solid Matrigel® dome supplemented with a specialized medium that mimics the native environment. With this culture system, the ISCs located within each crypt are capable of giving rise to the 3D organoid structure that mimics the adult intestine and contains all of the major cell types of the intestinal epithelium. Furthermore, intestinal organoids cultured in this system can be expanded, passaged and maintained indefinitely. IntestiCult™ Organoid Growth Medium (Mouse) is the first-ever complete and defined medium for generating intestinal organoids from mouse intestinal crypts. With a simple and easy-to-use protocol, IntestiCult™ is giving researchers an opportunity to conveniently incorporate organoids into their research.
References
- van der Flier LG and Clevers H. Annu Rev Physiol 71: 241-260, 2009
- Clevers H and Batlle E. Cell 152: 1198, 2013
- Barker N, et al. Genes Dev 22: 1856-1864, 2008
- Snippert HJ, et al. Cell 143: 134-144, 2010
- Buczacki SJA, et al. Nature 495: 65-69, 2013
- Sambuy Y, et al. Cell Biol Toxicol 21: 1-26, 2005
- Haegebarth A and Clevers H. Am J Pathol 174: 715-21, 2009
- Evans GS, et al. J Cell Sci 101: 219-231, 1992
- Fukamachi H. J Cell Sci 103: 511-519, 1992
- Ootani A, et al. Nat Med 15: 701-706, 2009
- Sato T, et al. Nature 459: 262-265, 2009
- Koo B-K, et al. Nat Methods 9: 81-83, 2012
- Sato T and Clevers H. Science 340: 1190-1194, 2013
- Leushacke M and Barker N. Gut 63: 1345-1354, 2014
- Sato T, et al. Nature 469: 415-418, 2011
- Farin HF, et al. J Exp Med 211: 1393-1405, 2014
- Grabinger T, et al. Cell Death Dis 5: e1228, 2014
- Wilson SS, et al. Mucosal Immunol, 2014
- Yui S, et al. Nat Med 18: 618-623, 2012
- Dekkers JF, et al. Nat Med 19: 939-945, 2013
- Schwank G, et al. Cell Stem Cell 13: 653-658, 2013
- Ranga A, et al. Adv Drug Deliv Rev 69-70: 19-28, 2014