Novel Hydrogels from AMSBIO for Advanced 3D Cell Culture
Three-dimensional (3D) cell culture models are growing in popularity and becoming preferred over conventional 2D cell culture because they can provide a more physiologically relevant platform to study complex biological processes across a wide range of research areas. Cells within 3D models, such as spheroids and organoids, experience cell-cell and cell-matrix interactions, nutrient and oxygen gradients, and a microenvironment that better recapitulates in vivo conditions. This ability to mimic the extracellular matrix (ECM) within human tissues and organs enhances the predictive power of 3D models for studying cellular behavior, tissue development, disease modeling, drug responses, and developing novel therapeutics.
Matrices play a crucial role in 3D cell culture, providing a necessary support structure or scaffold for cells to attach, grow, and interact within a 3D space. Hydrogels offer versatility in terms of their mechanical properties, degradability, and incorporation of bioactive molecules, making them popular for a wide range of applications.
What are Hydrogels?
Hydrogels are networks of polymers that have the unique ability to absorb water and undergo gelation. They are typically in a liquid state at 4⁰C but solidify into a gel when incubated at 37°C. This property allows hydrogels to encapsulate cells and support their growth within a 3D gel matrix while still allowing for the diffusion of nutrients, oxygen, and signaling molecules.
AMSBIO recently launched two new ECM hydrogel products that are optimized to support a wide variety of 3D cell culture techniques, including Patient-Derived Xenograft (PDX) studies as well as organoids and spheroids. Other ECM options are available from AMSBIO for induced pluripotent stem cell (iPSC) culture.
Extragel
Extragel is a hydrogel made from reconstituted basement membrane components, containing laminin, collagen IV, heparan sulfate proteoglycans, and a variety of growth factors. Extragel serves as a direct substitute for Matrigel™, Geltrex™, and Cultrex™ Basement Membrane Extract (BME), delivering comparable performance for applications involving organoid, induced pluripotent stem cells (iPSCs), and other advanced 3D cell cultures (Figure 1). Additionally, Extragel can be utilized for in vivo xenograft generation, making it suitable for translational PDX studies.
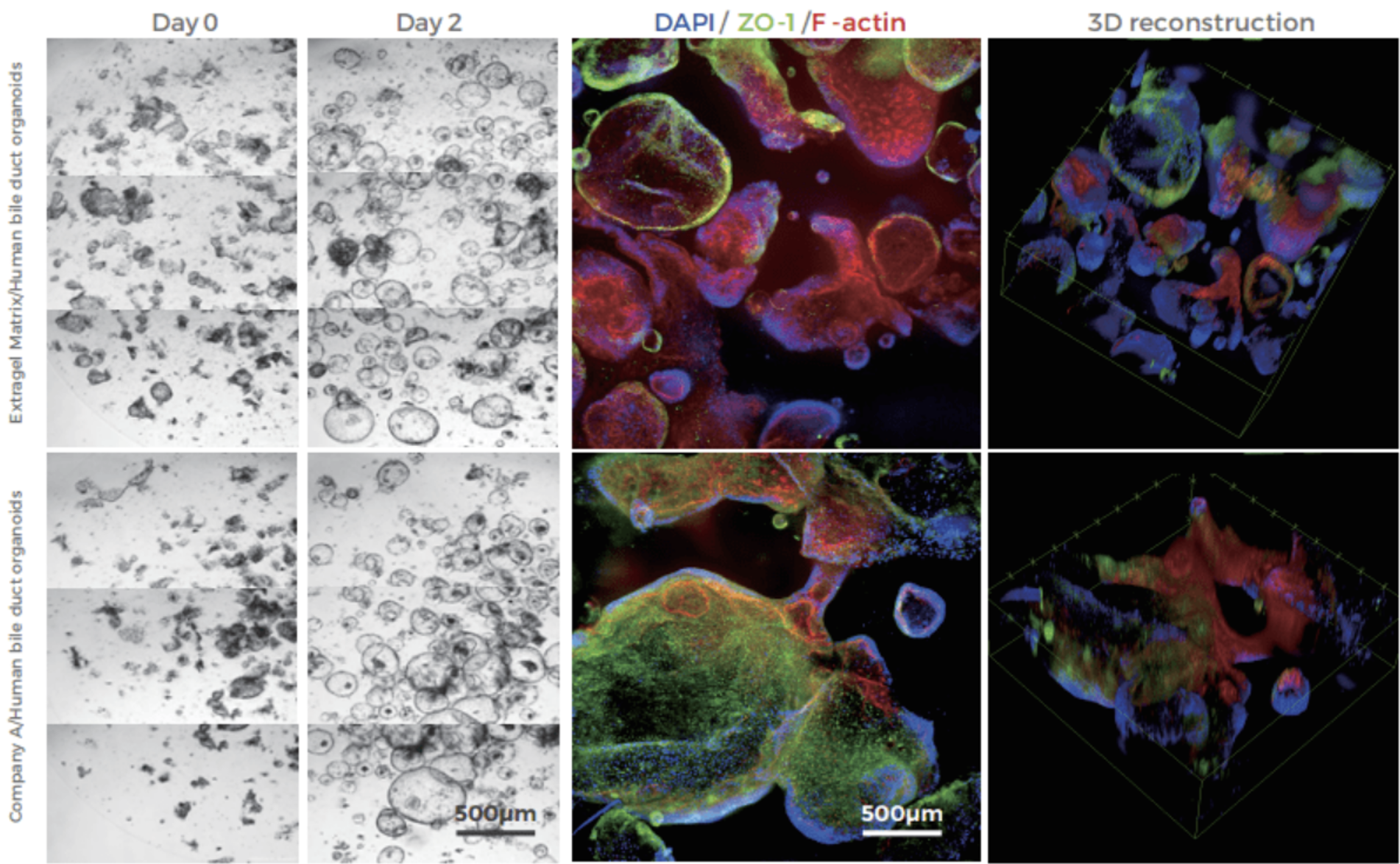
Known batch-to-batch variations in Matrigel™ composition and performance can introduce unwanted experimental variables and complicate data interpretation.Extragel’s superior lot-to-lot consistency because of stringent quality control allows for more reproducible experimental results. Extragel is LDEV-free, compatible with all cell culture media, and can be used either undiluted or diluted to achieve a specific protein concentration to support a wide range of 3D cell culture applications.
MatriMix Hydrogel
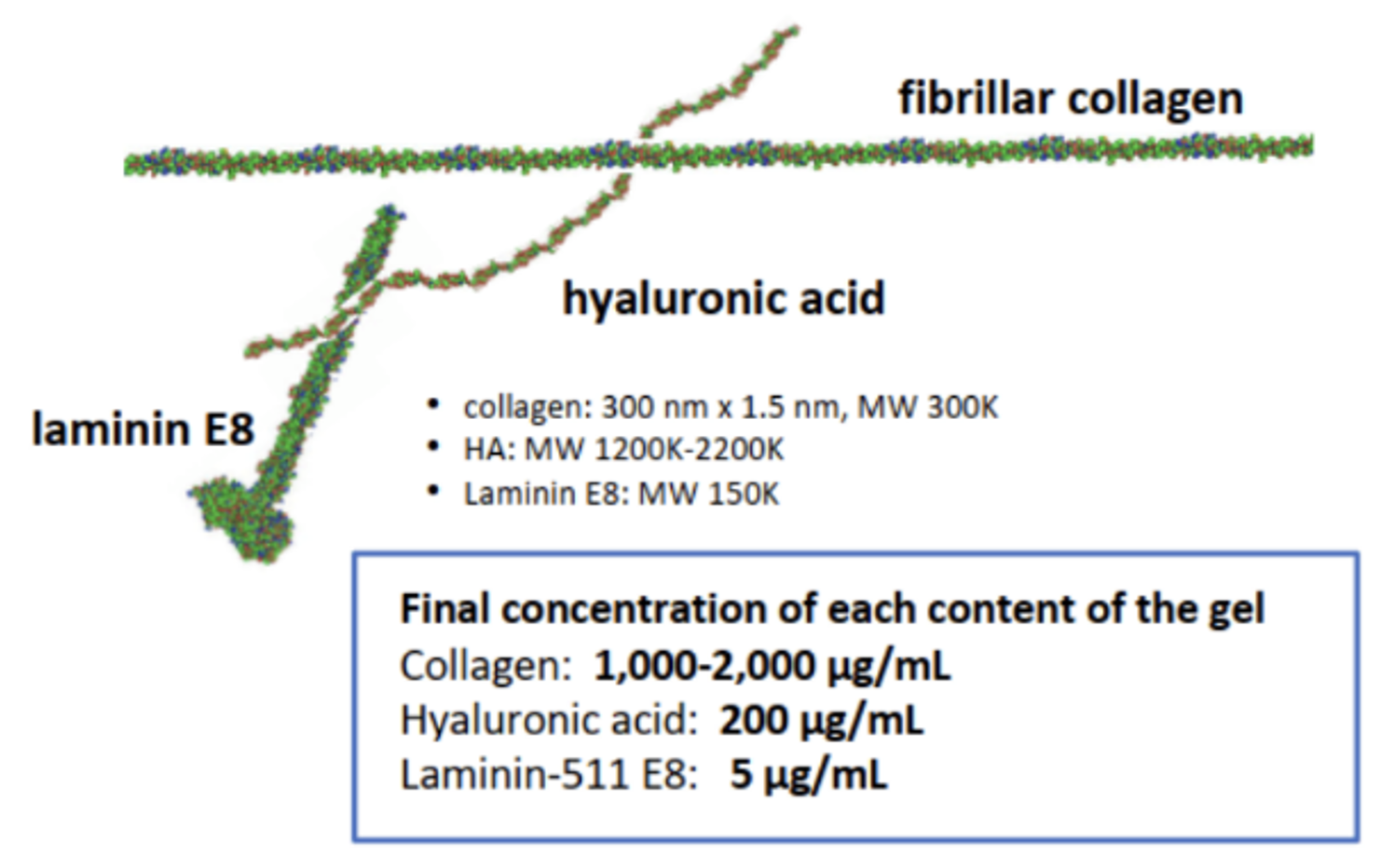
MatriMix Hydrogel is an innovative new 3D substrate that can provide more physiologically relevant and in vivo-like conditions compared to current alternatives. It is regulatory friendly and contains fully defined components including medical grade collagens, laminin-511 E8 fragments, and hyaluronic acid (Figure 2).
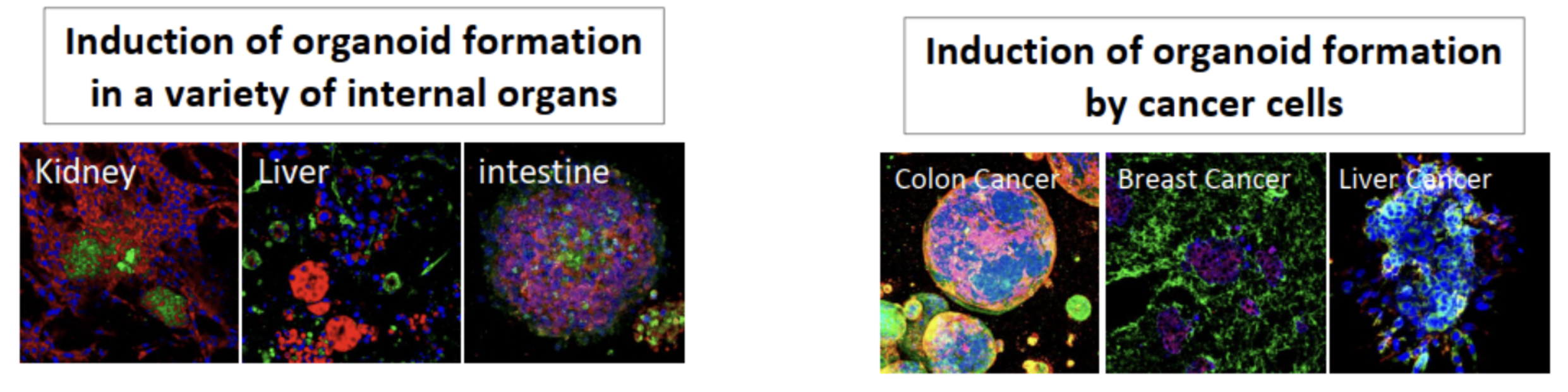
Extensive validation studies have confirmed that this hydrogel is a highly versatile substrate compatible with a wide range of cell types for the formation of organoids (Figure 3) and is well-suited for use in xenograft transplantation studies. Compared to Matrigel™, it is also a more biomimetic alternative for drug screening, delivering results that align better with in vivo testing without the need for transplantation into mouse models. Other benefits of MatriMix includes high batch-to-batch reproducibility and its ability to form a clear gel when incubated at 37°C, which enhances microscopic visualization during experiments.
Designed as a three-tube formulation kit, MatriMix offers unparalleled flexibility giving researchers the ability to customize the types and concentrations of individual components, tailoring the extracellular environment precisely to their target cells. The collagen type and concentration, laminin isoform, and various ECM components can be optimized to support the 3D culture of various organ tissues. The kit ships at a convenient 4 degrees and thus doesn’t need dry ice.
AMSBIO is a leading supplier of innovative products for life science research and is committed to developing innovative matrix solutions to serve the evolving needs of the scientific community.
For more information, please visit: https://www.amsbio.com/products/cells-cell-culture/3d-cell-culture-extracellular-matrices
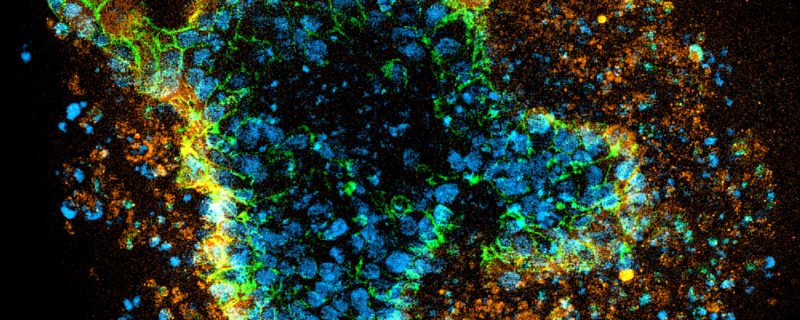
Header Image: “Patient-derived colorectal cancer organoids grown in Extragel. For the IF images, stains are DAPI in blue, LGR5 (intestinal stem cell marker- red or orange), and ECAD (epithelial cell marker located on the membranes – green). Image from Dr Ronan Mellin, Screen in 3D.”