Novel Stem Cell Technologies Presented at the 2014 Annual International Society for Stem Cell Research (ISSCR) Meeting
In June I attended the International Society for Stem Cell Research (ISSCR) in Vancouver and began a series of blogs related to the information presented at the meeting. In today’s blog I will be covering novel technologies for reprogramming and characterization, new cell lines available and stem cell manufacturing. The daily Innovation Showcases were particularity interesting and focused on new, state of the art products or approaches that enable stem cell culture and research.
Induction and Maintenance of a Novel State in Established Human Embyronic Stem Cell Lines
One Innovation Showcase highlight was a talk given by Dr. Huck-Hui Ng from the Genome Institute of Singapore on the “Induction of a Native State in Human Pluripotent Stem Cells.” Dr. Ng discussed his research on the induction and maintenance of a novel state, in established human embryonic stem (ES) cell lines, that more closely resembles the native pre-implantation epiblast state. Dr. Ng accomplished this by using mTeSR™1 medium and a combination of 3 small molecule inhibitors.
Dr. Ng describes how one of the goals of his lab is to map the biological networks that control the cell fate choices of stem cells. His research group began by comparing mouse and human embryonic stem cells. Interestingly, they observed that the morphology of mouse ES cells was distinct from that of human ES cells. In addition, the cytokines required to maintain pluripotency also differed, with mouse ES cells responding to LIF and BMP4 and human ES cells responding to FGF2 and activin.
Dr. Ng’s lab began looking for ways to shift human ES cells back to a state that is more similar to the native pre-implantation epiblast state. Initially, 11 different chemical inhibitors were applied to hESCs and had little impact on the cells, as the cells continued to differentiate. Next, different combinations of the chemical inhibitors were tested, and increased NANOG expression and pluripotent state used as indicators. A combination of 3 specific chemical inhibitors seemed to work. However, few cells survived after extended passaging, and those that did were unable to self-renew. By adding LIF to the three inhibitors, they obtained cells capable of self-renewal and expansion. With this combination Dr. Ng’s group were able to stably propagate these cells (3iL) for over 30 passages and maintain normal karyotype.
Characterization of the 3iL cells, demonstrated a re-wiring of the regulatory circuitry, as the 3iL transcriptome was distinct from hESCs and similar to that of epiblast cells. Thus, Dr. Ng and his group successfully pushed these hESCs into a cellular state that resembled the pre-implantation epiblast cells.
You can view the entire talk here:
Induction of a Native State in Human Pluripotent Stem Cells
Novel iPSCs Reprogramming and Differentiation Technologies
I was also fortunate to be able to see the Innovation Showcase “From Bland to Grand: iPSC Technology and its Applications,” presented by Dhruv Sareen, Regenerative Medicine, Cedars-Sinai Medical Center. Dr. Sareen walked through his group’s protocol and new efficiencies used in reprogramming iPSCs and also some of the applications they are researching with the use of iPSCs.
Dr. Sareen began the talk by discussing the protocol that they use for generating cell lines and some of the specific technologies they employ. One of the tools they use for characterization is the PluriTest gene chip characterization from Scripps to test for pluripotency. By using PluriTest, they were able to confirm close to 200 cell lines pass pluripotency with 85-87% passing and 15% needing further evaluation.
While they are able to confirm pluripotency with PluriTest, they were not able to predict how these lines would do when differentiated into different cell types and whether they would be able to differentiate into the three germ layers. So Dr. Sareen’s group employed the TaqMan hPSC scorecard panel and analysis software, which provides a score for each cell line on that line’s trilineage differentiation potential. It was suggested during the question and answer period that the use of the TaqMan hPSC scorecard panel could eliminate the need for teratoma assays.
Dr. Sareen gave a couple examples of how these cell lines were then provided to collaborators to use for specific research applications. Neural cells were given to a collaborator for use in generating a disease model and cardiac mesoderm organoids were provided to a collaborator to screen for cardiology drugs. Through these processes, Dr. Sareen’s group optimized their reprogramming by using high-content imaging and a defined media. They have generated greater than 85 cell lines that are available to investigators through the iPSC core at Cedars Sinai.
Dr. Sareen then discussed his own interests and applications for these cell lines in generating motor neurons to screen annotated chemical libraries looking for novel drug candidates. Dr. Sareen’s group has been able to generate 0.5 billion iMNs and are gearing up to screen 1,400-3,000 compounds.
You can view Dr. Sareen’s entire talk here:
“From Bland to Grand: iPSC Technology and its Applications,” presented by Dr. Dhruv Sareen
GMP iPSC Manufacturing
Lonza presented an Innovation Showcase titled “From the Nobel Prize to the Clinic: One Step Closer to iPSC-based Therapies,” in which Dr. Behnam Ahmadian Baghbaderani, Lonza Cell Therapy Development Services, outlined the process that Lonza has developed for manufacturing cGMP compliant human induced pluripotent stem cell (iPSC) master cell banks. One defining characteristic of these induced pluripotent stem cells is their ability to become any cell type in the body. It is thought that iPSC technology could then be used to develop cell transplantation therapies for the treatment of degenerative diseases such as diabetes, Parkinson’s disease and a number of cardiovascular diseases. The production of clinical grade iPSCs is considered a critical component to realizing the therapeutic potential of these unique stem cells.
In October 2012, the National Institutes of Health Center for Regenerative Medicine awarded Lonza a contract to produce GMP iPSC master cell bank. Due to the need for a robust, reproducible iPSC generation and banking process that complies with regulatory requirements, Lonza needed to develop and/or re-engineer what it refers to as the five key steps to generate clinical grade iPSCs:
Tissue acquisition – a donor program needed to be set up that meets FDA donor eligibility requirements
Cell isolation and priming – a xeno-free, defined method to isolate and culture the starting cell population prior to reprogramming
Reprogramming – an enhanced xeno-free, defined episomal plasmid-based reprogramming technology that utilizes the 4D-Nucleofector™ System
Expansion and banking – a xeno-free, defined culture system that supports the long-term growth and expansion of human pluripotent stem cells
Characterization and testing – purity, potency, identity and safety assays are needed to release the bank for clinical use
After more than three years of new technology development and process optimization, Lonza was ready for technology transfer of the process into manufacturing. During several training runs, Cell Therapy core technicians were trained on the process and standard operating procedures (SOPs) were updated. After the technicians had gained sufficient experience and understanding with the process, the next step was the engineering or validation runs. Here the technicians go into the GMP manufacturing suites and simulate running the process, with no support from the process development scientists, to prove they are able to successfully perform the process. At this point, the production SOPs are finalized. Assuming the results of these engineering runs meet specifications, then you are ready to start actual clinical grade, cGMP manufacturing. At the time of publication, manufacturing of the master cell bank is ongoing.
Lonza now offers cGMP manufacturing of iPSCs and their derivatives for Cell Therapy companies looking to outsource manufacturing of their cells for clinical development. Their comprehensive service offering allows clients to work with a single partner for tissue acquisition, process and assay development, media optimization and production, and cGMP manufacturing of therapeutic cells. The complete portfolio offers an attractive alternative to manufacturing in-house. Moreover, the media system – being commercialized as L7™ hPSC Culture System – is now available to basic and translational researchers alike.
Human Stem Cell Derived Skeletal Muscle Cells
Genea Biocells is an international stem cell company, headquartered in Australia that was displaying video time-lapse imaging of human stem cells being differentiated towards skeletal muscle. Please see video here – http://www.geneastemcells.com.au/Our-Products/Differentiated-Cell-Types/Skeletal-Muscle-Cells/cells
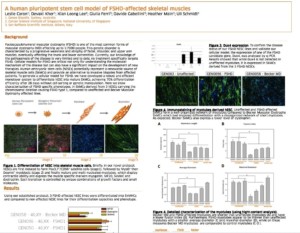
This custom protocol was developed in collaboration with a non-profit organization, FSHD Global, with the goals of generating a disease-in-a-dish model for Facioscapulohumeral Muscular Dystrophy (FSHD). Being the first such robust protocol of its kind, the video generated a great deal of interest, which was further enhanced by their research poster on the development of quantifiable traits using FSDH stem cell derived skeletal muscle cells. Please see poster below.
Besides custom differentiation protocols, Genea Biocells also offers automated high-content screening and assay development, a service they have provided to various biotechnology, pharmaceutical and academic groups. To complement this service, Biocells has also generated a bank of over 100 human embryonic stem cells (hESCs), representing over 30 distinct genetic disorders. The company is currently the largest contributor of NIH approved hESCs to the NIH registry and they are continuously expanding their catalog to better meet the needs of basic research and drug discovery.
GeneaBiocells_Poster ISSCR 2014-Final
ISSCR Series
This is just the second in a series of blogs covering ISSCR 2014. It was an excellent meeting and I highly recommend attending if you get the opportunity. Find out more about the ISSCR’s 2015 Annual Meeting in Stockholm, June 24-27 by visiting – ISSCR’s 2015 Annual Meeting
ISSCR Series Blogs
“Innovations Abound at the 2014 Annual International Society for Stem Cell Research (ISSCR) Meeting”
“Progress is Being Made Toward Using Cells as a Therapy for Type 1 Diabetes”