
Optimizing Vero Cell Culture Media with Animal Component-free, Recombinant Human Serum Albumin
Introduction
Vero cells are a monkey kidney cell line that has been a relative constant in the vaccine industry for over 3 decades. These cells have a long and successful history of safe vaccine production for many human and animal vaccines including polio, rotavirus, and smallpox. Vero cells are also a favored cell factory for the production of herpes, measles, and vaccinia-based viral vectors.
Vero cells are adherent cells that require a surface to attach to in order to proliferate and produce viruses. Production scale-up with adherent cells is generally more challenging than with suspension cell cultures due to the surface area and media formulation requirements. Vero cells grow well in a variety of vessels including flasks, cell factories, and microcarriers in bioreactors, however they must be supported in attachment and need a robust media to ensure good growth and productivity.
Traditionally, vero and other virus producing cells have been cultured in serum-containing medium. However the vaccine industry is moving toward defined, animal component-free media for vero cell culture and vaccine production, much like the move CHO cell-based manufacturing made several years ago. Using media formulated without serum or animal components reduces risk of contamination with adventitious agents, streamlines regulatory documentation, and can increase consistency in culture.
Human serum albumin in cell culture
Human serum albumin, has a multifaceted biological role and its inclusion in cell culture media has been shown to be extremely beneficial for the propagation of many cell types ex vivo. However, albumin isolated from human serum or bovine serum can still have variable performance and potential adventitious agent contamination risks.
Cellastim S – Recombinant human serum albumin
InVitria has introduced, Cellastim S, recombinant human serum albumin to address both the need for animal-component free media and the benefits that albumin provides in vero cell culture. Cellastim S is a lipid-rich recombinant human serum albumin used to enhance the production of numerous viruses via increased virus stabilization and enhanced Vero cell growth and production.
To ensure performance, every lot of InVitria’s Cellastim S is tested in cell culture. This minimizes the risks of lot-to-lot variability that can be seen in FBS or serum-derived albumin products.
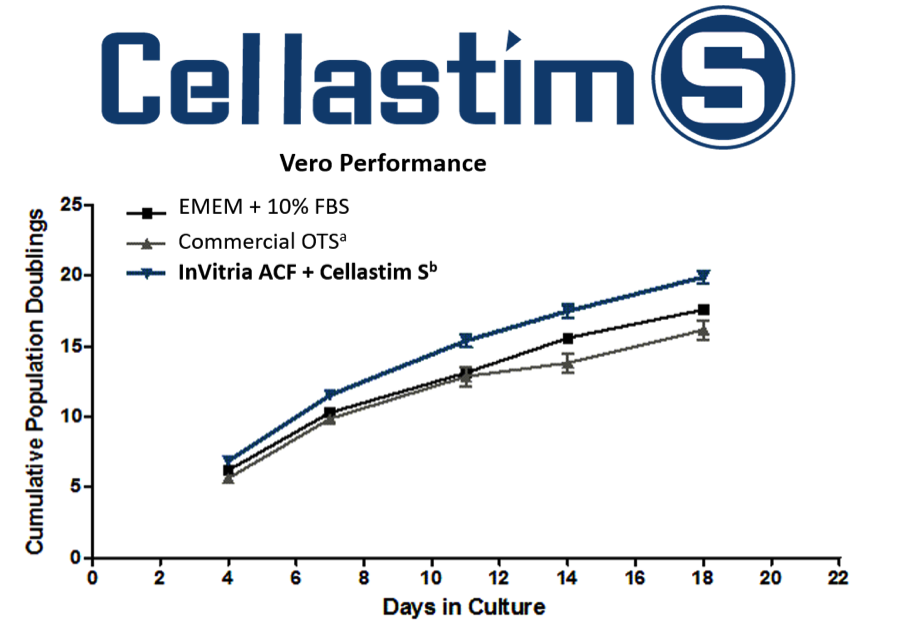
Figure 1 is a comparison between InVitria’s animal component free (ACF) Vero media containing Cellastim S and EMEM + 10% FBS and a commercial off-the-shelf (OTS) vero media. As you can see, the cells grown in media containing Cellastim S proliferate more rapidly than medias containing alternative sources of albumin.
aContains hydrolysates – Non-defined
bAnimal component free – Chemically defined
For more information and a complete list of Cellastim S cell lines and applications, please see – Cellastim S or feel free to Ask an Expert.