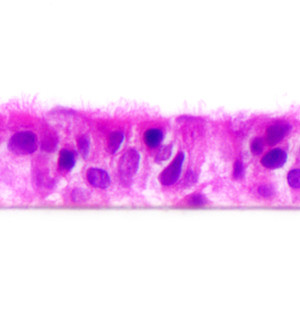
Producing 3D Tissue Models of the Airway Epithelium with PneumaCult™
The epithelial lining of the tracheobronchial region of the human respiratory tract is classified as a pseudostratified, ciliated columnar epithelium. Three cell types predominate in this region: ciliated cells, secretory cells (primarily mucus-secreting goblet cells) and basal cells.1 The tracheobronchial epithelium acts as a protective barrier that defends against a variety of inhaled insults such as toxins, pollutants and pathogens. Barrier function is maintained primarily as a result of three key characteristics of the epithelium:
- Tight junction proteins maintain the integrity of the epithelium and are critical to its function as a physical barrier.
- Secreted mucus traps particulate matter and pathogens, which are subsequently transported out of the airway by the coordinated action of ciliated cells in what is known as the mucociliary escalator.2,3
- Secreted protective mediators such as antimicrobial peptides and inflammatory mediators form a chemical and immunological barrier against pathogens and toxicants.4
There is a growing need for physiologically relevant models of the human airway epithelium, but recreating these complex functions in vitro is a considerable challenge. To appropriately model the in vivo human airway, specialized media and cell culture techniques have been developed that promote the formation of 3D structures that recapitulate the morphological and functional characteristics of the airway.
3D Tissue Models of the Airway Epithelium
Traditional submerged systems for 2D monolayer cultures can only support maintenance and expansion of basal cells. Under the culture conditions for 3D airway tissue models, however, basal cells can differentiate into a mucociliary pseudostratified epithelium containing ciliated, goblet and basal cells. In this in vitro cell culture environment, cells display beating cilia, mucus secretion, barrier properties and remodeling and restoration properties similar to the native airway epithelium. The two dominant 3D tissue models currently being used in the respiratory research field are air-liquid interface (ALI) cultures and sphere cultures.
Air-Liquid Interface Cultures for Modeling the Human Airway
The respiratory epithelium is exposed to the air in vivo, thus ALI cultures allow researchers to expand and differentiate epithelial cells in an environment that incorporates this physiologically relevant feature. In the ALI system, basal cells are seeded onto the semi-permeable membrane of a cell culture insert and submerged in a specialized liquid culture medium, such as PneumaCult™-Ex. Once the cells reach confluence, they are “air-lifted” by removing the medium from the upper chamber of the insert, exposing the apical surface of the cells to the air (view the video protocol).
Figure 1. Differentiate Airway Epithelial Cells into a Pseudostratified Mucociliary Epithelium with PneumaCult™
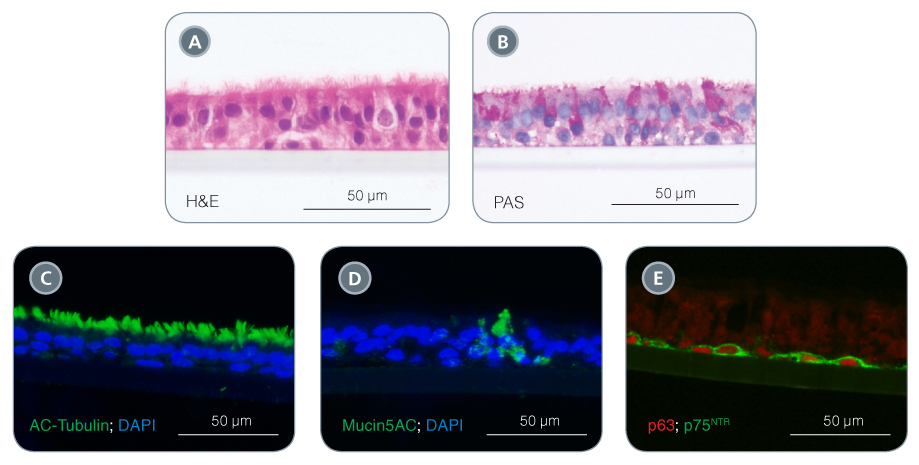
When using PneumaCult™-ALI, this process triggers extensive differentiation to produce a pseudostratified mucociliary epithelium (Figure 1). In addition to containing mucus-secreting and ciliated cell types, the ALI cultures also retain physiological epithelial barrier function by forming intercellular tight junctions and demonstrating high transepithelial electrical resistance. ALI cultures have been noted as being the most physiologically relevant in vitro model for studying the airway epithelium.5 This system allows researchers to study cell biology and infection, culture patient-derived cells to model diseases, and test the effects of aerosolized particles (including drug formulations and cigarette smoke) on the respiratory epithelium.
Sphere Cultures for High-Throughput Studies
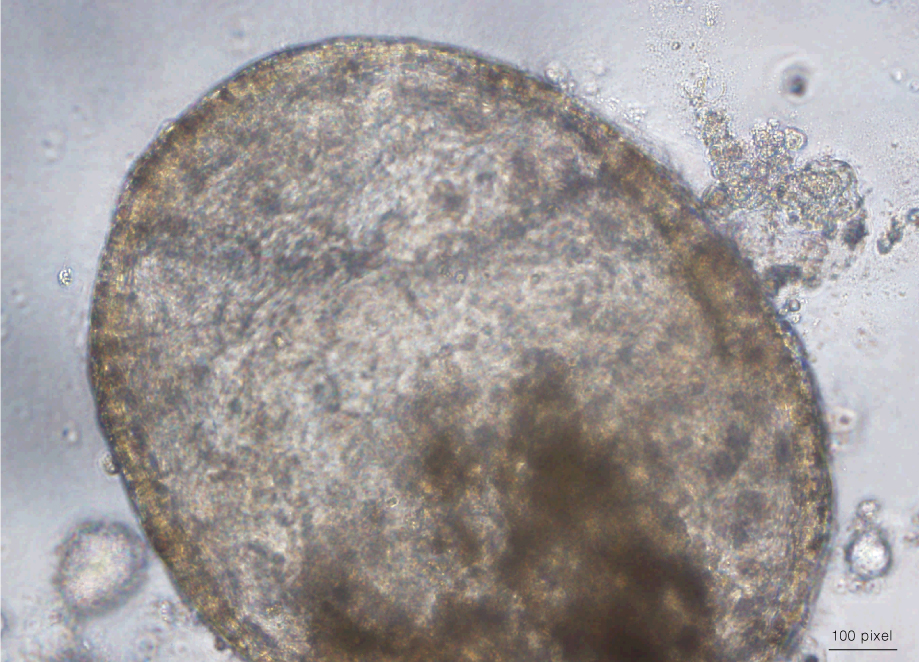
For experiments that require a culture method that is easily adaptable to a high-throughput format, 3D sphere cultures provide an alternative to ALI cultures. Airway epithelial cells can be cultured as spheres by suspending the cells in PneumaCult™-ALI and plating them on a tissue culture vessel coated with 40% Corning® Matrigel® / PneumaCult™-ALI (view the full protocol here). The cells arrange into spheres, differentiate and form a pseudostratified mucociliary epithelium with the apical surface of the cells pointing into the lumen. Video 1 shows a bronchosphere cultured in PneumaCult™-ALI exhibiting these features, including coordinated beating of cilia.
Video 1. Bronchosphere Culture in PneumaCult™-ALI
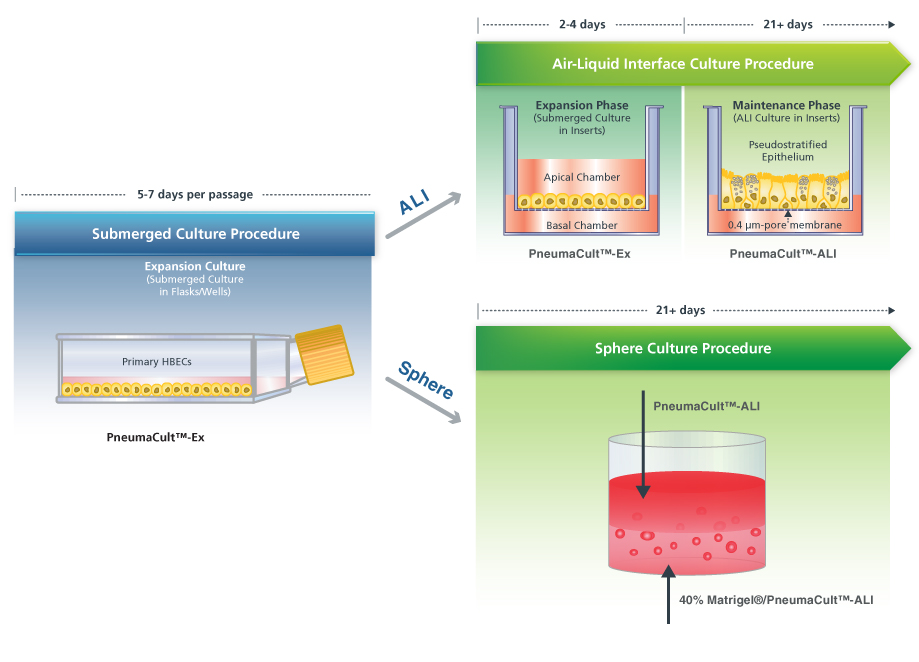
The PneumaCult™ Culture System
PneumaCult™-Ex and PneumaCult™-ALI are defined media for airway epithelial cells and do not contain bovine pituitary extract (BPE) (Figure 2). PneumaCult™-Ex is designed for expansion of airway cells in submerged culture, and PneumaCult™-ALI is specialized to allow for extensive differentiation of airway cells in ALI or sphere cultures. By using a defined and BPE-free culture system, researchers can reduce experimental variability and maximize reproducibility (view data). Learn more about the PneumaCult™ system here. If you have questions about how you can start using ALI or sphere cultures in your respiratory research, please contact us at pneumacult@stemcell.com.
Figure 2. Overview of the PneumaCult™ Culture System
Footnotes
-
1. Ehrhardt C et al. (2008) In Vitro Models of the Tracheo-Bronchial Epithelium. In: Ehrhardt C & Kim K (Eds.) Drug Absorption Studies (pp. 235-57). USA: Springer US.
-
2. Fahy JV & Dickey BF (2010) New Engl J Med 363(23): 2233-47.
-
3. Button B et al. (2012) Science 337(6097): 937-41.
-
4. Tam A et al. (2011) Ther Adv Respir Dis 5(4): 255-73.
-
5. Prytherch Z et al. (2011) Macromol Biosci 11(11): 1467-77.