
Understanding FBS Alternatives – Frequently Asked Questions
The ability to culture mammalian cells in vitro has been a vital scientific tool for decades and is used for a range of life sciences applications, from fundamental research to translational medicine. Fetal bovine serum (FBS) is the most widely used cell culture media supplement and has been an effective, broad-spectrum additive to support the growth of a range of cell types since the 1950s. FBS is a complex mixture of components, such as serum albumin, hormones, vitamins, trace elements, growth factors and other undefined constituents that support cell attachment, proliferation, and maintenance for many different cell types. FBS can also protect cells from large pH shifts by increasing the buffering capacity of the media and mitigates the physical cellular damage from shear forces during culture agitation.
However, because FBS is a by-product of the meat industry, the global supply is dictated and impacted by many factors, including consumption levels, feed prices, and environmental and pathogen-related issues like drought and disease. Challenges in availability combined with higher demand for FBS over time and increased ethical concerns surrounding its use has led researchers to invest a great deal of time and effort to find an ideal alternative to replace FBS in cell culture.
We had the opportunity to ask cell culture supplement experts Jeremy Amberson, Chief Operating Officer and Hal Shearin, Research and Product Development at RMBIO about this paradigm shift away from FBS and discuss the specifics of their FBS alternative, Fetalgro EX®. Our questions and their answers are presented below:
What are the biggest challenges with using FBS?
While FBS is attractive as a cell culture supplement because of its broad utility for a range of primary cells and cell lines, there are some significant challenges to overcome. The primary challenge with using FBS is the cost; it’s the most expensive cell culture media supplement available and prices constantly fluctuate. Another challenge for FBS from a scientific perspective is that it is an undefined biological material with batch-to-batch variation in quality and composition. Variability in the approximately 1,800 proteins and over 4,000 metabolites in FBS can cause inconsistencies in cell culture performance that affect experimental reproducibility. Additional issues around the biosafety and ethics of using FBS in cell culture highlight the importance of identifying a sustainable FBS alternative.
When would it be advantageous to use an FBS alternative instead of FBS?
In our discussions here, we will focus on FBS alternatives that have a broad compatibility across cell types, so-called ‘generalist’ media additives, as opposed to cell type specific FBS replacements. The primary advantage of general FBS alternatives is the cost. General FBS replacement products are less expensive than FBS yet can deliver similar results. Comparable efficacy at a fraction of the cost makes FBS replacements ideal in situations where budget is a concern and in cases where the specific nutritional requirements of a particular cell type/line have not been established, making a universal non-FBS supplement desirable.
Why not switch from FBS to a serum-free or chemically defined formulation?
Serum-free media and chemically defined media are specialized formulations typically designed to meet the nutritional needs of one cell type/line. The high cost and specificity of these defined formulations may not be suitable for researchers depending on their end goals. General FBS replacements help fill the gap between FBS and chemically-defined serum-free formulations because they are less restrictive, exhibit reduced variability, and can support the growth of a wide variety of cell types at a fraction of the cost of FBS.
What is Fetalgro EX®?
Fetalgro EX® is a high-performance, cost-saving, serum-based general FBS alternative developed by RMBIO to reduce the burden of the increasing cost and variable supply of FBS. The proprietary formula is supplemented with defined additives to support the culture of many different cell types.
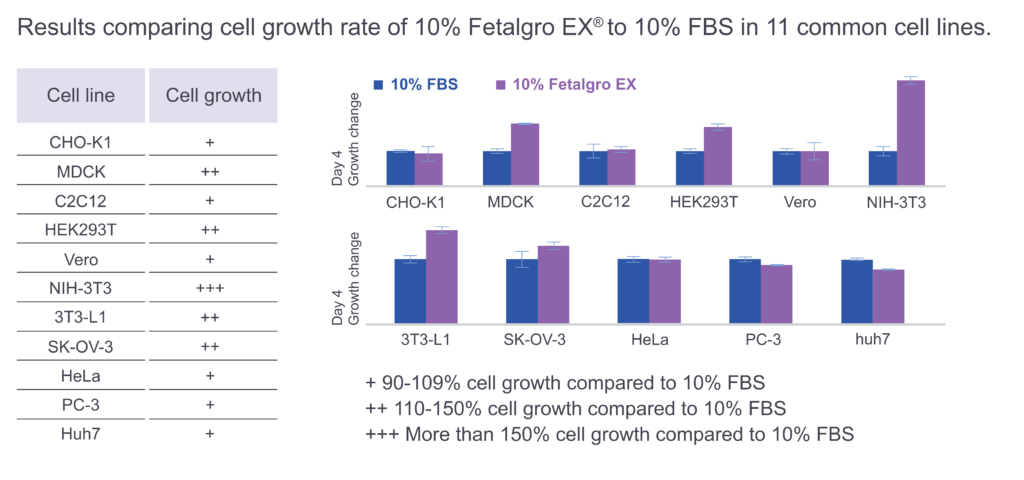 Is it necessary to adapt cells grown in FBS to media containing Fetalgro EX®?
Is it necessary to adapt cells grown in FBS to media containing Fetalgro EX®?
No, Fetalgro EX® can be applied directly to cells previously cultured with FBS without adaptation.
Can Fetalgro EX® be substituted 1:1 with FBS in my media?
Yes, Fetalgro EX® is a direct replacement for FBS at the same concentration. For example, 10% FBS can be substituted with 10% Fetalgro EX® in culture media.
What cell types are compatible with Fetalgro EX®?
Fetalgro EX® works well with a variety of cell types including epithelial and epithelial-like cells, fibroblasts, myocytes and myoblasts, adipocytes and adipocyte-like cells, hepatocytes, cancer cells, and recombinant cell lines suitable for a wide range of applications in manufacturing, research, and development. These include but are not limited to CHO, HEK293, VERO and HeLa cells.
Are there any differences observed with respect to cell morphology when cells are grown in Fetalgro EX® compared to FBS?
Fetalgro EX® showed comparable morphology across multiple cell types when compared to FBS.
How does Fetalgro EX® compare to FBS with respect to safety and specifications?
Fetalgro EX® offers the same rigorous quality controls as common FBS products:
- Raw materials are sourced from qualified, USDA-audited suppliers.
- All raw serum used at RMBIO is certified by the International Serum Industry Association (ISIA) to be sourced in accordance with their strict guidelines.
- Fetalgro EX® meets European Union requirements for technical blood products and is eligible for export to the EU.
- All RMBIO products are produced in a GMP compliant and FDA registered facility.
- Fetalgro EX® features low endotoxin and hemoglobin levels.
- Fetalgro EX® undergoes mycoplasma, viral, and sterility testing.
The advantages of FBS alternatives like Fetalgro EX® are numerous and address some of the key challenges associated with FBS faced by the scientific community. Overcoming the variability and cost of FBS offers a more sustainable approach to support cell culture practices now and in the future.
For more information, please see Fetalgro EX®
About our Experts
 Jeremy Amberson, Chief Operating Officer at RMBIO
Jeremy Amberson, Chief Operating Officer at RMBIO
As COO and former manager of R&D, Jeremy harnesses skills at the nexus of science, engineering, and business management to develop, manufacture, and market high-quality and innovative products for the research, biopharma, and clinical diagnostics industries. In addition to being a veteran of the United States Air Force, Jeremy has held various positions and businesses in the fields of environmental remediation, ecological research and restoration, and biotechnology for the past 25+ years. Jeremy holds degrees in philosophy, biology, and an MS in ecology from the University of Montana.
 Hal Shearin, Research and Product Development at RMBIO
Hal Shearin, Research and Product Development at RMBIO
Hal has over two decades of experience in tissue culture, recombinant and molecular biology, neuroscience, and animal research, including several scientific co-authorships. Upon graduating from St. Mary’ College of Maryland, a National Public Honors College, with a BA in Biology, he began his career as a lead primary-cell culture technician at Lonza Bioscience in Walkersville, MD. Since, he has conducted research in and managed several labs in Montana, including at Montana State University, before joining the team at RMBIO.