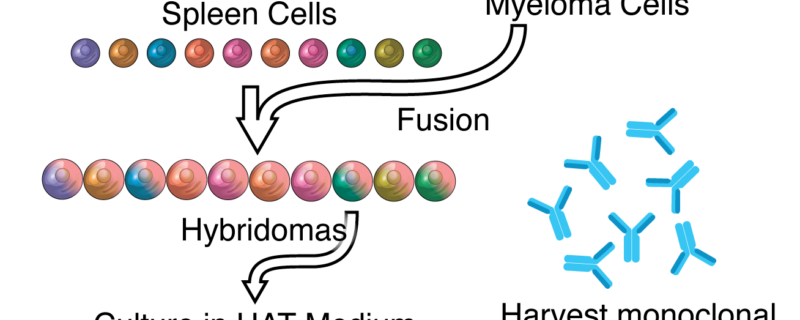
In Celebration of Hybridoma Cell Culture
In recent months we have provided in depth coverage on hybridoma cell culture and it continues to be a highly searched topic at The Cell Culture Dish. In light of its popularity, we wanted to provide a brief history of the use of hybridoma cells and a review of our cell culture coverage that includes new technologies, best practices and key cell culture recommendations. According to a report late last year issued by PhRMA, there are over 900 biotech drugs in development in the United States, of these 300 are monoclonal antibodies. While the use of hybridoma cells has certainly evolved since its first use to create monoclonal antibodies in the 1970’s, it is important to recognize that it is still being used today over 35 years later. Hybridoma technology changed the future of medicine and continues to have a major impact on monoclonal antibody development today.
Hybridoma technology was a result of needing to move production of antibodies from the human body to the lab and this concept was heavily researched in the early 1970’s. In 1975 two researchers, Georges Kohler and Caesar Milstein, joined together and discovered the technology for producing a monoclonal antibody using hybridoma technology. In another laboratory around the same time, Niels Jerne had discovered a plaque assay to show a visual representation of antibody producing B cells. These three researchers changed medicine forever and went on to share the Nobel Prize in medicine in in 1984. Because Milstein and Kohler never patented their discovery, it opened the door for new monoclonal antibody medicines to be discovered over the next thirty years.
Ever since hybridoma technology was discovered in the 1970’s, scientists have been improving the technology. They have worked to enhance the antibodies themselves by improving the way they are accepted by the human body and reducing side effects. Through genetic engineering, scientists have been able to move away from a final product that is full mouse antibody, to a chimeric antibody (about 33% mouse and 67% human) and now some companies are manufacturing humanized antibodies (generally 5-10% mouse and 90-95% human). In addition to improving the genetic make-up of the antibody, work is continually being done to improve manufacturing techniques and antibody yield. These topics have been covered in the Cell Culture Dish blogs listed below.
Recent Blogs that Address Hybridoma Cell Culture:
- Innovations in Hybridoma Media
- Serum Free Hybridoma Culture – Key Recommendations
- Strategies for Improving Antibody Production in Hybridoma Cells
- Utilizing Automated Clone Selection to Improve Antibody Production in Hybridoma Cells
- Advancing Media to Improve Antibody Production in Hybridoma Cells
- Using Bioreactors to Improve Antibody Production in Hybridoma Cells
- Alternatives to Fetal Bovine Serum in Cell Culture Media – Challenges and Perspectives
- Combining Insulin and Transferrin in Cell Culture Supplements