Corning® Nu-Serum™ I/IV Growth Medium Supplements for Reduced-serum Conditions with Fibroblast/Kidney Epithelial Cells
Introduction
With the movement towards reduced-serum and serum-free conditions for cell culture, defined formulations and low-protein alternatives are of increasing value. Fetal bovine serum (FBS) is traditionally used as a supplement in media for cell and tissue culture at final concentrations of 5% to 10%. However, it can introduce risk as FBS lot-to-lot variation may negatively impact cellular and molecular studies, and can introduce artifacts due to the presence of unknown compounds, proteins, growth factors, and other undefined components1. Additionally, the high protein content of serum can introduce complications with protein purification. Corning® Nu-Serum™ growth medium supplements provide more cost-effective, low protein alternatives that can be utilized as a 1:1 direct replacement for FBS in cell culture media.
In the first arm of this study, MRC-5 (human lung fibroblasts), were cultured in media containing either 10% FBS or 10% Corning Nu-Serum I to assess supplement performance. In the second arm, Vero cells (African green monkey kidney epithelial cells) were grown in media supplemented with FBS, or in media with Corning Nu-Serum IV. Cell morphology, doubling time, and viability over five consecutive passages were monitored. When used as a 1:1 direct replacement for FBS in cell culture media, Nu-Serum I and IV yielded stable cell doubling times and good cell viability that were comparable to the FBS-containing media controls.
Materials/Methods
MRC-5 cells (ATCC® CCL-171™) and Vero cells (ATCC® CCL-81™) were thawed into Minimum Essential Media (MEM, Corning Cat. No. 10-010-CVR) containing 10% FBS (Corning Cat. No. 35-010-CV). Evaluation of media with either FBS, Nu-Serum I (Corning Cat. No. 355500) for MRC-5 cells, or Nu-Serum IV (Corning Cat. No. 355504) for Vero cells was initiated in Tissue Culture (TC)-treated T-25 flasks at passages 4-12. For each study, two T-25 vessels per condition were seeded with MRC-5 cells at 7.5×103-1.5×104 cells/cm² or Vero cells at 3×103-5×103 cells/cm² in 0.3 mL/cm² of MEM containing 10% final concentration of either FBS, Nu-Serum I, or Nu-Serum IV, respectively. MRC-5 and Vero cells were cultured for 72 to 96 hours, depending on initial seeding density, at 37°c in 5% CO2 in a humidified cell culture incubator. Cultures were monitored daily for cell morphology and confluence using an EVOS® FL Imaging System. Cells were harvested based on the confluence and morphology of the control flasks with media containing 10% FBS, regardless of the confluence of the flasks with Nu-Serum supplements. Cells were harvested with Accutase® cell detachment solution (Corning Cat. No. 25-058-CI) and centrifuged at 250 x g for 5 minutes. The supernatant was aspirated, and cells from replicate flasks were re-suspended in their appropriate media and pooled together for cell enumeration and viability measurements using a NucleoCounter® NC-200™ automated cell counter (ChemoMetec), and subsequent passaging. Each study continued for five sequential passages and was repeated three independent times.
Results and Discussion
To assess the ability of Corning Nu-Serum I and Nu-Serum IV growth medium supplement to directly replace FBS in cultures, the addition of 10% final concentration of Nu-Serum I/IV in MEM was compared to 10% FBS in MEM for five consecutive passages of fibroblast/kidney epithelial cell culture using MRC-5 and Vero cells. Cell morphology and confluence was monitored daily, as well as cell counts and viable cell densities on the day of each passage.
During the course of the study, typical MRC-5 fibroblastic morphology was observed with media containing Nu-Serum I serum supplement, as directly compared to the FBS containing media control (Figure 1a). Typical Vero kidney epithelial morphology was observed with media containing Nu-Serum IV serum supplement being directly compared to the FBS-containing control medium (Figure 1b). MRC-5 and Vero cell confluence appeared similar between flasks with media containing 10% FBS, Nu-Serum I, and Nu-Serum IV serum supplements (Figure 1).
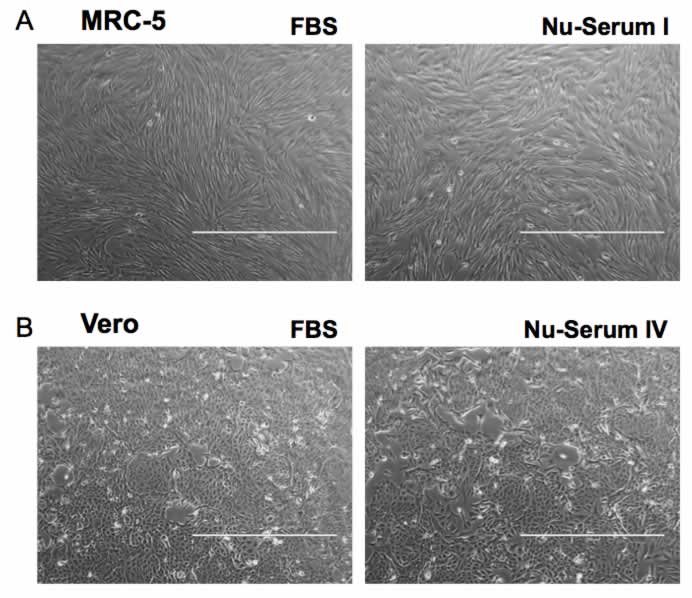
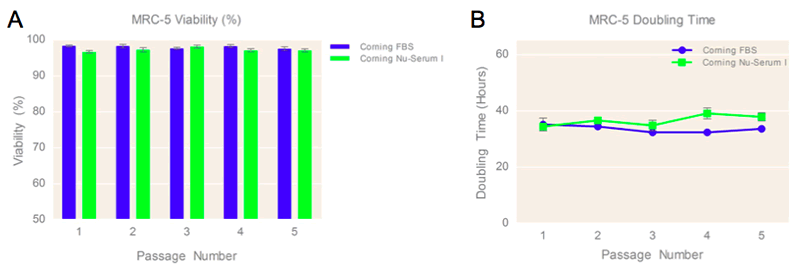
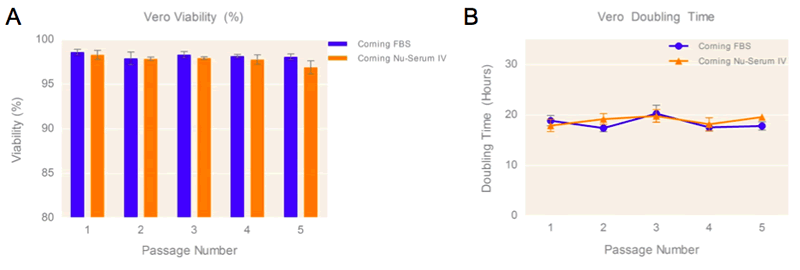
MRC-5 cells cultured in media containing 10% Corning® Nu-Serum™ I, and Nu-Serum IV respectively for Vero culture displayed similar cell yields to cells cultured in medium supplemented with 10% FBS, and high-cell viability (>96%) over the course of five passages (Figures 2a, 3a). The cultured cells in media containing 10% Nu-Serum supplements displayed comparable cell doubling times (Figure 2b, 3b) over the course of five passages, suggesting positive cell proliferation rates and no sign of strenuous growth conditions when using this media supplement. The similar cell doubling times also support visual assessment of high cell confluence of MRC-5 and Vero cells cultured using Nu-Serum I & IV supplements when compared to FBS-containing media control.
Conclusions
Corning® Nu-Serum I/IV can be used as a 1:1 direct replacement for FBS in cell culture media for culturing fibroblast/kidney epithelial cells, such as MRC-5 and Vero respectively, with similar cell doubling time and high cell viability.
A Guest Blog by Christopher Bortz & Hilary Sherman, Corning Incorporated, Life Sciences, Kennebunk, ME
Footnotes
-
1. Gstraunthaler G, Lindl T, and van der Valk J. (2013). A plea to reduce or replace fetal bovine serum in cell culture media. Cytotechnology, 65(5):791-793. http://doi.org/10.1007/s10616-013-9633-8.