Development of DoE based fed-batch strategies for high-producing CHO cell cultures
Successful optimization of Chinese hamster ovary (CHO) cell culture for biomanufacturing has a significant impact on productivity, protein quality, manufacturing efficiency and economic feasibility. To achieve the highest performing cell culture, a fed-batch culture strategy is frequently used. In fed-batch culture, it is important to begin with a robust base media and then add a complimentary feed strategy.
There are a number of tools that are used to inform decisions about feed strategy. One of the most beneficial is to utilize Design of Experiments (DoE) to test the performance of media with various feed supplements at different concentrations. A good DoE can help determine not only which feeds are best, but also concentration and timing for adding feed components.
Many current platform processes feed a fixed-volume bolus of concentrated supplements at scheduled intervals. However, recent developments show that there may be more effective methods for scheduling feeding. A more dynamic feed strategy that responds to the needs of the culture rather than feeding on a set static schedule could yield better results.
In a recent poster, “Development of DoE based fed-batch strategies for high-producing CHO cell cultures“, presented by GE Healthcare and the Vienna Institute of BioTechnology, the authors developed and tested the benefit of a dynamic feed strategy that was responsive to the nutrient needs of a mAb-producing recombinant CHO cell line. The utilization of DoE in the development of this strategy along with corresponding data is presented in the poster. I have presented some poster highlights in this article.
Study Design and Results
To begin, the authors employed DoE using MODDE™ statistical software in batch culture to select the best supplements. Eight HyClone™ Cell Boost™ supplements were added to HyClone CDM4NS0 medium. In this evaluation authors only selected the Cell Boost supplements that were beneficial for the overall culture performance. Non-performing Cell Boost supplements were excluded from further studies.
Next the authors fine-tuned the feed ratio of selected supplements in fed-batch culture. Using a DoE approach, the selected Cell Boost supplements were added daily to the cultures at different ratios. Daily feed additions to replenish consumed nutrients substantially improved mAb and peak cell concentrations as well as viable cumulative cell days (VCCD) compared with batch cultivation. Using this information, feed ratio of the selected supplements was optimized.
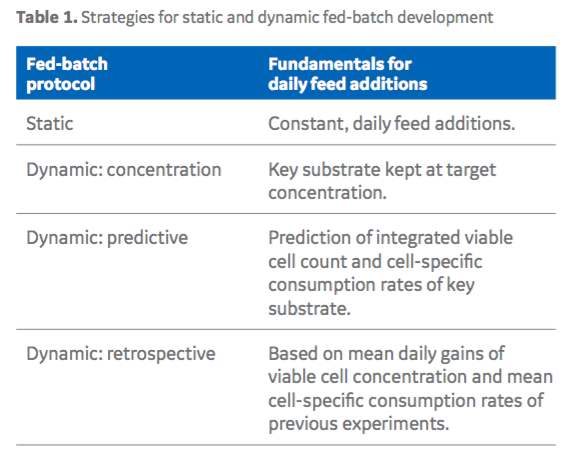
Then they looked at a constant vs. dynamic feed schedule in fed-batch culture. In most fed-batch protocols feed is typically added at a set amount on set days, but this can lead to problems with over or underfeeding culture. To improve on this approach, authors looked to optimize feeding strategies by looking at three different dynamic approaches using the optimized Cell Boost feed ratios (Table 1).
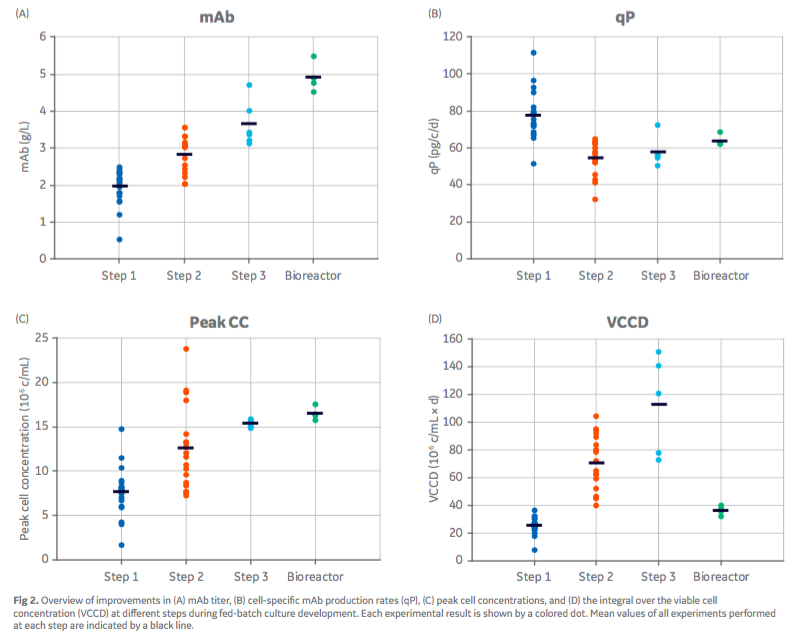
The most promising approaches were investigated using automated bioreactor runs under controlled conditions. While, bioreactor culture conditions slightly improved mAb titers over shake flask culture, the dynamic retrospective feed strategy yielded 10% higher titers than the constant strategy. Figure 2 from the poster, gives an overview of the improvements made in different steps during development of a fed-batch culture process.
Conclusions
The established methodology for fed-batch culture development is a rapid protocol to select well-performing feed supplements and optimize their ratio to the culture requirements. In three steps, mAb titers were boosted 2.5× from 1.9 g/L to 4.9 g/L. Product glycosylation and charge variants could be influenced by the newly selected basal media and feeds compared with a legacy fed-batch process. The amount of aggregated product was not altered.
Additional Reading on Feed Design Strategies:
“Fed-batch culture – Optimizing feed strategies now and in the future,” The Cell Culture Dish
“Transcriptome analysis reveals strategies for CHO cell culture media design and feed-spiking strategy to improve batch culture,” The Cell Culture Dish