
Improving lab productivity and reproducibility by taking a fresh look at existing workflows and protocols
Podcast: Download (Duration: 24:17 — 33.4MB)
Subscribe Here: Apple Podcasts | Spotify | RSS | More
Subscribe to the Cell Culture Dish Podcast on: iTunes | Google Play
Show Notes
We began the interview by discussing Beckman Coulter’s Spring Cleaning Methods Infographic, which depicts key areas of lab operation that could benefit from a review to improve operational productivity and reproducibility.
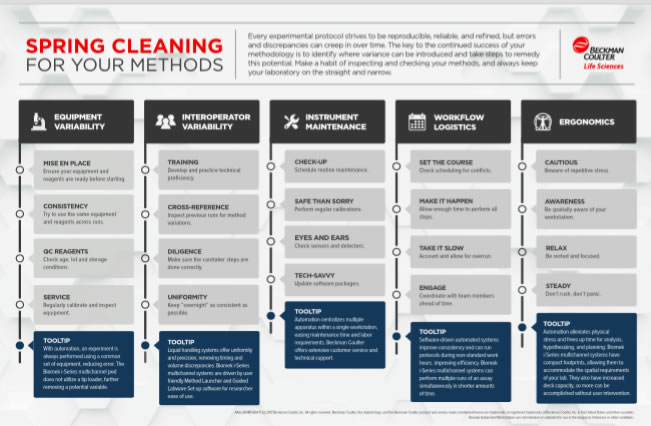
I asked Sarah of the operations listed in the infographic, which did she feel was most difficult for labs to maintain. She felt that the biggest challenge was in minimizing variance between operators. She explained that even if operators don’t realize it, there are subtle differences in the ways we carry out tasks that make it difficult to have absolute consistency and control of operations. Timing also plays a role, the time of the day in which a task is carried out can affect how an operator conducts that task. Thus even with the same operator there can be variability. When analyzing results it is always important to think about how the assay is being performed and to try to limit the impact of any variations.
Next, I asked Sarah about the Spring Cleaning Methods Article and how protocol refinement can be used to maximize time and resources. Sarah described that automation can go a long way here and that the biggest benefit of adding automation is the ability to achieve consistency. With automation, you take out the operator variation because the equipment is going to run the same process regardless of the time of day or who operates it.
Another benefit to automation is the ability to set up an operation based on what is best for the assay, not on what is the best in terms of lab operating hours. For instance, Sarah shares that incubation is often set up overnight for 16 hours or so because 16 hours is about the amount of time that people are out of the lab overnight. However, this may not actually be the most ideal amount of time for incubation, is it better with less? Because automation isn’t’ dependent on someone being present in the lab it is possible to refine protocols and possibly build a more robust assay by evaluating what is really the best incubation time.
I then brought up the importance of reviewing reproducibility in the lab and looking for ways to enhance this. We discussed that again operator variability plays a big role in reproducible results. I asked Sarah what she thought were the primary challenges and possible solutions. She reiterated that we are all human and do things in a slightly different way and training new operators on a specific task can also be an area for review. Sarah provides a great example of this. She says, it’s like teaching a child to ride a bike, you know how to ride a bike because you’ve done it so long that it becomes second nature, now imagine trying to train someone who has never ridden a bike how to do it exactly the way you do it. There are certain principles you can explain but getting them to replicate your way exactly is a tough task. In the case of pipetting, things like how fast you let your finger off the plunger, whether you condition your tips, etc. are differences that are hard to even identify as differences, but they can impact consistency. Not to mention transferring these protocols to a collaborator’s lab. These little differences can add up to larger variations.
Next we discussed challenges to reproducibility due to variations in equipment use. Sarah shared that different readers, pipetors, liquid handling systems, etc. can also all run slightly different based on the model, the brand and the age of the equipment. This can lead to differences in assay results. Ways to combat this include regular equipment maintenance and keeping equipment clean by wiping up spills and splashes that can impede operation over time. Periodic maintenance on schedule is important and should include inspection and parts replacement when necessary. She went on to say that many companies run performance qualifications and she encourages this be conducted on a scheduled basis.
We then talked about ergonomics and how examining methods can improve productivity and reduce injury. Things like bench height, how you are sitting in relation to the bench, and how far you have to move between pieces of equipment are examples of areas where ergonomic adjustment can make a difference. It is important to have a clear path during an experiment and to be able to be efficient in your movements. Moving items around in an established lab can be difficult, but starting a new assay can be a good time to evaluate any possible changes you could make to improve the ergonomics. You also have to consider where the equipment is located with respect to other pieces of equipment. Equipment that vibrates or has high heat generation can affect nearby equipment and can have an impact on results.
Lastly, I asked Sarah if she had anything else she would like to add. She said that bringing in new equipment provides an opportunity to save time and limit waste in running assays by providing a faster process. However automation does not always guarantee consistency and that if there are outdated protocols still available on the equipment that could be run in error, this can result in variation. It is important to always clean up old or outdated protocols, the goal is for it to be easy for an operator to walk up and select the right protocol.