New Wave: Cell-Based Therapies
Regenerative medicine is beginning to change the market for both pharmaceuticals and medical devices. Utilizing the new technology of cell-based therapies has the potential to impact prolonged medical conditions such as vascular applications. The use of these live cells as a mechanism for self-repair with the potential to cure a disease has peaked the interests of companies to invest in this market.
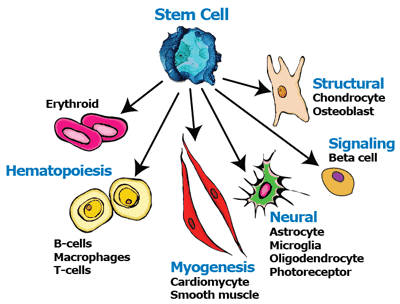
Stem cells have become the hot topic in developing this new opportunity in the market. However, cultivating these live cells can present many challenges from cell differentiation, inconsistency, and contamination. Maintenance of the undifferentiated state of the stem cells while propagating the cells is a challenge. The manipulation of the media formulation has to accomplish the culturing of the cells in their targeted state, whether partially differentiated or undifferentiated until the cells are ready to be administered to the target tissue/organ.
Many media formulations that are currently being utilized in research and development studies still have animal-derived components in them. Regulations are not enforced to remove the animal components until after clinical trials. Then, the challenge is to maintain the same performance without the animal components that the cells are adapted to grow in.
To be successful, scientists need to be able to manufacture these cell therapies, using an animal component free system, without compromising the performance of the cells as a therapeutic.