How a Next Generation Proteomics Platform is Accelerating the Fight Against COVID-19
Olink provides a high throughput, cost-effective proteomics platform for screening ~1500 protein biomarkers using a minimal volume of sample.
Since the beginning of the COVID-19 pandemic, researchers from around the world have been working tirelessly to understand the biological pathways of the disease and develop effective therapeutics. One important enabling approach is proteomics. Proteomics is broadly defined as the study of proteins. A proteome is an entire set of proteins produced in an organism during its life cycle or at a given moment in time. In medicine, proteomics is used to provide insight into health and disease and increase our understanding of biology in real-time.
Proteomics for COVID-19 Research
When analyzing a new disease, like COVID-19, it is crucial to understand the underlying biological pathways and biomarkers present in the disease state. This information allows researchers to identify potential diagnostic and therapeutic approaches. In a global pandemic situation, this data needs to be generated both at a large scale and as quickly as possible. What would normally be investigated over several years now needs to be identified in a matter of weeks.
An open hypothesis approach is ideal in this situation because it allows researchers to rapidly study the plasma proteome to look for correlations between protein biomarkers and disease. However, the challenge with this approach is the throughput needed to investigate such a large number of proteins
Olink’s proteomics portfolio does just that, providing high throughput proteomics solutions to the research community. This results in rapid screening and identification of biomarkers associated with disease state, the ability to determine prognosis severity, and study the immunological response, among others. Olink’s technology, when coupled with analysis of related comorbidities, allows the assessment of multiple risk factors for severe COVID-19 and provides tools to monitor patient disease progression and identify potential new drug targets.
Olink Proteomics – Next Generation Proteomics
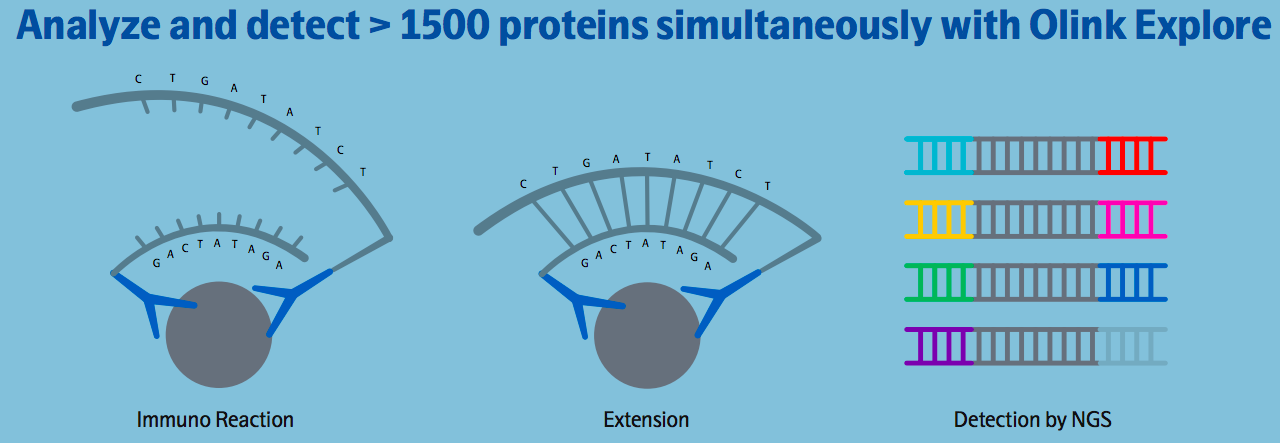
Olink uses Proximity Extension Assay (PEA) technology, which combines an antibody-based immunoassay with a digital readout using either quantitative real-time PCR (qPCR) or Next Generation Sequencing (NGS). The unique attributes of this technology support a scalable, multiplex, and highly specific approach where the concentration of more than a thousand protein biomarkers can be quantified simultaneously.
Proximity Extension Assay
PEA is a dual-recognition immunoassay, where two matched antibodies labeled with unique DNA oligonucleotides simultaneously bind to a target protein in solution. This brings the two antibodies into proximity, allowing their DNA oligonucleotides to hybridize, which serves as a template for a DNA polymerase-dependent extension step. The resulting double-stranded DNA “barcode” is unique to each specific antigen and quantitatively proportional to the initial concentration of the target protein. Hybridization and extension are immediately followed by PCR amplification. This provides a highly scalable technique with exceptional specificity. Because the PEA assay requires less than one drop of blood to measure the protein profile in a patient sample, there is no need to worry about applications where samples are precious but scarce.
Low Abundant Plasma Proteome
There is increasing interest in tapping into the information held within the low abundant plasma proteome. Most proteins show a large variation in abundance between individuals. Some proteins are at such minute concentrations that they are barely detectable, however, it is this low abundant plasma proteome where the richest store of knowledge related to dynamic biological changes in health and disease may be found. The OlinkÒ Explore 1536 is well suited to investigate proteins found in the low abundant plasma proteome by providing a substantial increase in throughput and detection for a significantly expanded protein library in a cost-efficient way.
This type of research is extremely valuable in COVID-19 research as the low abundant proteins often reveal the most surprising and valuable insights for understanding biological pathways and disease.
Olink® Explore 1536
The Olink Explore 1536 platform couples PEA with Next Generation Sequencing (NGS) to enable the readout of 1536 assays in 96 samples simultaneously using just 3 µL of sample. It is perfect for studies involving large numbers of human serum or plasma samples where high throughput technology is required. The 1536 assays are arranged into four 384-plex panels that run in parallel, generating over 130,000 data points in less than 36 hours and over 1.3 million protein measurements per system per week. It also has a broad dynamic range with assays that span ten logs (fg/mL – mg/mL).
Scalability
Olink’s PEA technology can achieve a high level of multiplexing while maintaining exceptional data quality. Unlike many other immunoassay formats, Olink’s panels are designed such that no antibody cross-reactivity occurs during the multiplexed assay. Olink spans small to large scale studies, without the need to change platforms, providing a smooth transition between large-scale screening studies and validation of the specific protein signatures that have been identified.
Quality and validation
The Olink Proteomics assays are rigorously quality controlled, and the validation data is completely transparent and freely available. For more information on the analytical performance of the Target 96 panels and validation data on sensitivity, dynamic range, specificity, precision, and scalability, please see Transparent Data Validation.
COVID-19 Research Studies using Olink Proteomics
Olink’s protein biomarker panels are currently being used in a significant number of on-going and planned COVID-19 studies worldwide. These studies cover a wide range of research topics including:
- Understanding basic biology and prognosis of disease severity
- Patient stratification by disease severity
- Longitudinal sampling (to monitor disease progression and recovery)
- Prediction of cytokine release storms in COVID-19 patients
- Prediction/prevention of ARDS (Acute Respiratory Distress Syndrome)
- Outcome predictions in cardiovascular disease patients with COVID-19
Use of protein biomarkers to determine COVID-19 severity and outcome
In one of the largest longitudinal COVID-19 studies to date, Olink and Massachusetts General Hospital (MGH) collaborated to execute one of the first major protein biomarker studies ever done for COVID-19 using the new Olink Explore 1536 platform. The platform allowed close to 1500 proteins to be rapidly analyzed in a cohort of around 400 COVID-19 patients and symptomatic control subjects. The study generated over 1.3 million protein data points along with associated clinical parameters that were shared with the wider scientific community. This data can be used to augment other researchers’ findings and accelerate the development of new therapies as well as a better understanding of the underpinnings of COVID-19.
This was the largest COVID-19 proteomics study to date and required the powerful technology and throughput that the Olink Explore 1536 provides. The Olink Explore 1536 was able to screen for almost 1500 biomarkers across multiple time points using just a small blood sample. What took the Olink Explore 1536 one to two weeks, would have taken months using mass spectrometry technology. As a result, researchers identified proteins that may predict the severity and outcome of COVID-19 infections in patients at the time of hospital admission. In application, this could be used early on in the disease state to monitor and prioritize patients that are at risk. It could also lead to future clinical trial intervention and tailored therapies based on these results.
Other studies on protein biomarkers using Olink Proteomics have also provided vital information about COVID-19. The Department of Internal Medicine, Radboud UMC, Nijmegen, Netherlands, reported how they used targeted proteomics with Olink panels to investigate almost 270 proteins and discovered novel and unexpected pathophysiological pathways involved in COVID-19 infection. These discoveries advance understanding of the disease and may provide starting points for future therapies.
In another example, immune response in mild versus severe COVID-19 infections was investigated in a study published in Science and led by researchers at Stanford University. Multiplex analysis of cytokines in plasma was used to assess the immune response of COVID-19 patients with mild-to-severe disease from two geographically distant cohorts. Study results revealed mechanisms and potential therapeutic targets for COVID-19.
Comorbidities
Another key area of research is in the relationship between COVID-19 and associated comorbidities, identified by the World Health Organization (WHO) as being associated with worse health outcomes. In a recent study in The Lancet, Olink’s technology was used in a large epidemiological study investigating the association of plasma ACE2 levels with COVID-19 risk, as ACE2 is a SARS-CoV-2 virus cellular receptor. Specifically, Olink’s assay for ACE2 was used to measure ACE2 plasma levels in more than 10,000 subjects. The authors found clear associations between plasma levels of ACE2 and the risk of major cardiovascular events, such as heart failure, stroke, and myocardial infarction.
Future COVID-19 Studies
Even with the promise of vaccines and recombinant antibody therapies on the way, much work still needs to be done. To date, the cellular mechanisms underlying the immune response and disease severity of COVID-19 are poorly understood and a comprehensive overview of the inflammation processes of COVID-19 is still lacking. The pathophysiology behind COVID-19 must be explored further with deep profiling of the inflammatory system. Understanding and predicting disease severity is critical in improving patient outcomes. Lastly, the identification of new therapeutic targets and a greater understanding of the therapeutic impacts on multiple organ systems and inflammatory biomarkers is a pressing need.
Non Covid-19 Applications
In addition to the ways that Olink Proteomics is being used in COVID-19 research, it is also being implemented across other disease areas, including cardiovascular disease, cancer, inflammation, neurology, autoimmune disease research, etc… Proteins are being used to identify biomarker signatures that predict patients at higher risk of various disease outcomes.
Pharmaceutical companies are also using Olink Proteomics to discover new drug targets, and with the ability to screen thousands of biomarkers, have a higher likelihood of success. This work informs how disease biology works, why we develop disease in the first place, and why some patients respond favorably to a drug while others do not.
Drug Discovery Consortium
To further work related to drug discovery, Olink is a central technology for use by the SCALLOP consortium. SCALLOP is a collaborative framework of academic and industry members for the discovery and follow-up of genetic associations to proteins on the Olink Proteomics platform. They aim to identify novel molecular connections and protein biomarkers that are causal in disease. So far, 25 principal investigators from 20 research institutions have joined the effort, which now comprises summary-level data on SNP to protein level associations from almost 65,000 patients or controls. SCALLOP welcomes new members.
Summary
Olink Proteomics offers two platforms to meet the needs of researchers working on COVID-19 research and in other disease areas. The Olink Explore 1536 assay is perfect for high throughput screening studies with large sample numbers. The Olink Target 96 range of panels are great for more targeted investigations around specific diseases or biological processes using one or several panels that are most relevant to the subject of study.
With both platforms, Olink technology enables proteomic research that previously could not be conducted. For instance, scientists are now able to search for new, actionable protein signatures within the low abundant plasma proteome that can be applied to liquid biopsy investigations to improve disease detection, aid more personalized healthcare, and allow a better understanding of real-time human biology.
Olink is providing a substantially reduced price on their services for COVID-19 related studies to support the crucial work that will help better understand and eventually better treat the disease. They are committed to help accelerate the gathering and publication of essential data required to deal with the current global pandemic.
For more information, please see the infographic above as well as:
Related articles:
Proteogenomic Strategies to accelerate Drug Discovery and Increase Success
Next Generation Proteomics is the Missing Piece in Completing the Precision Medicine Puzzle