
The Dish’s Weekly Biotechnology News Wrap Up – December 1, 2023
This week’s headlines include: Amgen expands pact with Amazon to usher drug manufacturing into the AI era, WHO authorizes emergency use of Novavax’s updated COVID shot, Thermo Fisher teams up with Flagship Pioneering to forge new life science tool startups, Lilly Puts Potential $660M on the Line for Protein-Protein Interaction Blockers, 2023’s Fiercest Women in Life Sciences, Valneva says EU regulator accepts its chikungunya vaccine application, and Stakeholders urge FDA to retain interchangeability statements on biosimilar labeling.
Podcasts:
Check out our podcast channel. We have over 100 great podcasts covering drug discovery, stem cell culture, upstream and downstream biomanufacturing and more! Click below to download from iTunes or Google play:
![]()
![]()
In Case You Missed It, Recent Articles on Cell Culture Dish and Downstream Column:
![]()
Expand Productivity in Upstream Bioprocessing with the XCell® ATF System
 The XCell® ATF System from Repligen is an innovative technology that consists of a controller and a cell retention device that enables the intensified cell culture to exponentially boost productivity in upstream bioprocessing. Upstream process intensification with the XCell ATF System increases the output of existing facilities resulting in higher productivity, lower costs, lower risks, increased flexibility, and faster production…
The XCell® ATF System from Repligen is an innovative technology that consists of a controller and a cell retention device that enables the intensified cell culture to exponentially boost productivity in upstream bioprocessing. Upstream process intensification with the XCell ATF System increases the output of existing facilities resulting in higher productivity, lower costs, lower risks, increased flexibility, and faster production…
Enhancing Biopharmaceutical Production: The Impact of Chemically Defined Peptides on Biopharma Upstream Process Efficiency and Cost
Cell culture has become an essential part of biopharmaceutical manufacturing for the production of recombinant proteins,  antibodies, vaccines, and gene therapies for a range of therapeutic indications. As a result, cell culture media formulations have evolved significantly to meet the needs of the industry. In particular, today’s formulations must be chemically defined and cannot contain animal-derived components. This can be achieved by replacing complex components with amino acids, vitamins, and other ingredients. The shift towards non-animal-derived formulations is driven by the overall need to increase product safety, reproducibility, and regulatory compliance. In addition, as production cell lines become more specialized, there is a growing need for customizable media formulations and higher nutrient concentrations to increase productivity by intensifying and optimizing biopharmaceutical processes, especially in monoclonal antibody production platforms…
antibodies, vaccines, and gene therapies for a range of therapeutic indications. As a result, cell culture media formulations have evolved significantly to meet the needs of the industry. In particular, today’s formulations must be chemically defined and cannot contain animal-derived components. This can be achieved by replacing complex components with amino acids, vitamins, and other ingredients. The shift towards non-animal-derived formulations is driven by the overall need to increase product safety, reproducibility, and regulatory compliance. In addition, as production cell lines become more specialized, there is a growing need for customizable media formulations and higher nutrient concentrations to increase productivity by intensifying and optimizing biopharmaceutical processes, especially in monoclonal antibody production platforms…
Novel Hydrogels from AMSBIO for Advanced 3D Cell Culture
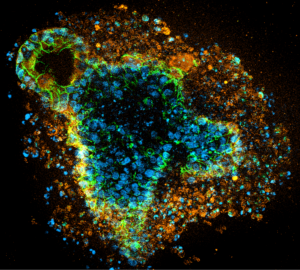 Three-dimensional (3D) cell culture models are growing in popularity and becoming preferred over conventional 2D cell culture because they can provide a more physiologically relevant platform to study complex biological processes across a wide range of research areas. Cells within 3D models, such as spheroids and organoids, experience cell-cell and cell-matrix interactions, nutrient and oxygen gradients, and a microenvironment that better recapitulates in vivo conditions. This ability to mimic the extracellular matrix (ECM) within human tissues and organs enhances the predictive power of 3D models for studying cellular behavior, tissue development, disease modeling, drug responses, and developing novel therapeutics. Matrices play a crucial role in 3D cell culture, providing a necessary support structure or scaffold for cells to attach, grow, and interact within a 3D space. Hydrogels offer versatility in terms of their mechanical properties, degradability, and incorporation of bioactive molecules, making them popular for a wide range of applications…
Three-dimensional (3D) cell culture models are growing in popularity and becoming preferred over conventional 2D cell culture because they can provide a more physiologically relevant platform to study complex biological processes across a wide range of research areas. Cells within 3D models, such as spheroids and organoids, experience cell-cell and cell-matrix interactions, nutrient and oxygen gradients, and a microenvironment that better recapitulates in vivo conditions. This ability to mimic the extracellular matrix (ECM) within human tissues and organs enhances the predictive power of 3D models for studying cellular behavior, tissue development, disease modeling, drug responses, and developing novel therapeutics. Matrices play a crucial role in 3D cell culture, providing a necessary support structure or scaffold for cells to attach, grow, and interact within a 3D space. Hydrogels offer versatility in terms of their mechanical properties, degradability, and incorporation of bioactive molecules, making them popular for a wide range of applications…
Cell and Gene Therapy Advancements, Challenges, and the Path to Commercial Success
Cell Culture Dish, in collaboration with a panel of gene therapy experts, published two e-book publications in 2018 and 2020 that covered all aspects of gene therapy commercialization in depth. As the field continues to broaden and mature, we wanted to provide an updated look—with insight from industry experts—at the current landscape across therapeutic targets, manufacturing processes, and regulatory frameworks and how progress in these areas are propelling cell and gene therapies forward. Although there is huge promise, the field is still young and there are many challenges that persist. Here we explore the obstacles that remain on the path to commercial success and highlight the key opportunities for future advancement and sustainability…
covered all aspects of gene therapy commercialization in depth. As the field continues to broaden and mature, we wanted to provide an updated look—with insight from industry experts—at the current landscape across therapeutic targets, manufacturing processes, and regulatory frameworks and how progress in these areas are propelling cell and gene therapies forward. Although there is huge promise, the field is still young and there are many challenges that persist. Here we explore the obstacles that remain on the path to commercial success and highlight the key opportunities for future advancement and sustainability…
Overcoming the Challenges of Scaling Stem Cell Cultures for Allogeneic Therapies
 In this Ask the Expert session, we spoke with Whitney Wilson and Antonio Fernandez-Perez, both Field Application Scientists with Corning Life Sciences, about the incredible potential of allogeneic therapeutics, ways to solve the current challenges they face, and what the future may hold…
In this Ask the Expert session, we spoke with Whitney Wilson and Antonio Fernandez-Perez, both Field Application Scientists with Corning Life Sciences, about the incredible potential of allogeneic therapeutics, ways to solve the current challenges they face, and what the future may hold…![]()
f(x) Columns for Mechanistic Chromatography Modeling
Mechanistic modeling is an approach that uses computer simulations based on known physiochemical phenomena to understand  and describe a variety of chromatographic modes. These simulations can effectively decrease the number of physical experiments that need to be performed during downstream process development to optimize parameters that can affect chromatographic performance. Advantages of mechanistic modeling include time savings, improved process understanding and robustness, and support of regulatory decision making…
and describe a variety of chromatographic modes. These simulations can effectively decrease the number of physical experiments that need to be performed during downstream process development to optimize parameters that can affect chromatographic performance. Advantages of mechanistic modeling include time savings, improved process understanding and robustness, and support of regulatory decision making…
ÄKTA ready™ Chromatography Systems from Cytiva
 In the current landscape of biopharmaceuticals, single-use technologies are being increasingly incorporated into bioprocessing workflows. They offer several benefits such as enhanced manufacturing flexibility, reduced risk of contamination, and decreased downtime that can streamline process scale-up and manufacturing. These benefits align with the goals of improving efficiency, product safety, and accelerating time-to-market for novel therapeutics while being adaptable to the evolving needs of the industry…
In the current landscape of biopharmaceuticals, single-use technologies are being increasingly incorporated into bioprocessing workflows. They offer several benefits such as enhanced manufacturing flexibility, reduced risk of contamination, and decreased downtime that can streamline process scale-up and manufacturing. These benefits align with the goals of improving efficiency, product safety, and accelerating time-to-market for novel therapeutics while being adaptable to the evolving needs of the industry…
Enhance Downstream Bioprocessing Efficiency with AKBA’s Award-Winning MOTIV™ Inline Buffer Formulation (IBF™) Systems
Buffer formulation is an essential part of downstream bioprocessing to produce safe and effective biopharmaceuticals more  efficiently. Buffers are aqueous solutions with precise pH and ionic strength that can stabilize proteins, aid purification steps, as well as regulate enzymatic activity – and are essential to creating optimal conditions for various biochemical reactions. Specific buffer compositions are required throughout the downstream process, often in volumes that can reach thousands of liters…
efficiently. Buffers are aqueous solutions with precise pH and ionic strength that can stabilize proteins, aid purification steps, as well as regulate enzymatic activity – and are essential to creating optimal conditions for various biochemical reactions. Specific buffer compositions are required throughout the downstream process, often in volumes that can reach thousands of liters…
Cytiva MabSelect™ VL protein L affinity resin
 Monoclonal antibodies (mAbs) represent a rapidly growing class of therapeutics designed to target and bind to specific proteins to treat a variety of diseases, including cancer, autoimmune disorders, and infectious diseases. They can be custom engineered to have specific properties that suit specific therapeutic needs, such as increased potency, longer half-life in the body, or reduced immunogenicity. As the field continues to advance, the molecular diversity of mAbs is increasing rapidly, where non-traditional mAb formats such as bispecific antibodies, fragments, and antibody-drug conjugates now account for up to a quarter of projects in the clinical pipeline. Conventional purification protocols relying on protein A may not always be suitable for these novel antibody variants since they may require new ligands or base matrices for effective capture of target molecule and separation of impurities. Therefore, the development of chromatography resins that can accommodate these variants is crucial for manufacturing and process development in the evolving landscape of mAb therapeutics…
Monoclonal antibodies (mAbs) represent a rapidly growing class of therapeutics designed to target and bind to specific proteins to treat a variety of diseases, including cancer, autoimmune disorders, and infectious diseases. They can be custom engineered to have specific properties that suit specific therapeutic needs, such as increased potency, longer half-life in the body, or reduced immunogenicity. As the field continues to advance, the molecular diversity of mAbs is increasing rapidly, where non-traditional mAb formats such as bispecific antibodies, fragments, and antibody-drug conjugates now account for up to a quarter of projects in the clinical pipeline. Conventional purification protocols relying on protein A may not always be suitable for these novel antibody variants since they may require new ligands or base matrices for effective capture of target molecule and separation of impurities. Therefore, the development of chromatography resins that can accommodate these variants is crucial for manufacturing and process development in the evolving landscape of mAb therapeutics…
Cool Tool – GoSilico™ Chromatography Modeling Software from Cytiva
Biopharmaceutical manufacturers are continuously striving to find efficiencies and make improvements across downstream  processing steps to improve product quality, reduce costs, and accelerate the development of safe and effective biopharmaceuticals. Chromatography plays a critical role in the purification of biopharmaceutical products. However, developing chromatography processes can be a challenging task because it involves balancing factors such as selectivity, yield, and productivity while maintaining the purity and quality of the final product. Strategies for process development are evolving and include design of experiments (DoE) and high throughput process development (HTPD). These strategies accelerate process development and increase process understanding, but they require multiple experiments and extensive data analysis, which can be time consuming and resource intensive…
processing steps to improve product quality, reduce costs, and accelerate the development of safe and effective biopharmaceuticals. Chromatography plays a critical role in the purification of biopharmaceutical products. However, developing chromatography processes can be a challenging task because it involves balancing factors such as selectivity, yield, and productivity while maintaining the purity and quality of the final product. Strategies for process development are evolving and include design of experiments (DoE) and high throughput process development (HTPD). These strategies accelerate process development and increase process understanding, but they require multiple experiments and extensive data analysis, which can be time consuming and resource intensive…
Simplified, Closed Slurry Preparation to Manage a Wide Range of Chromatography Media for DAC LC Columns

Slurry preparation is a critical step in preparing a well-packed column and achieving a successful bioprocess purification. However, this step can be challenging as different therapeutics require different chromatography media to achieve effective chromatographic separation. For instance, therapeutics in the 5 to 15 kDa range, such as insulins and oligonucleotides utilize rigid and spherical chromatography, which can be more difficult to work with. These media are based on a wide range of matrices including silica gel, polystyrene-divinylbenzene, polymethacrylate, agarose and hydroxyapatite, each with its own packing properties. As these processes are scaled up to larger column diameters, packing media becomes more complex and more crucial for success…
White Paper:
Physiologix™ as a Serum Replacement Supplement to Promote the Growth and Performance of Primary T Cells – Download White Paper
 Support T cell growth, expansion, transduction efficiency, and viability
Support T cell growth, expansion, transduction efficiency, and viability
Identifying a T cell culture media that can maximize cell performance and seamlessly transition from development to commercialization is complex. Physiologix is a GMP and xeno-free serum replacement that can drive desirable critical quality attributes at superior levels to FBS and human serum.
As an alternative to serum, Physiologix also helps avoid regulatory hurdles and optimizes development.
In this white paper, we review a comprehensive study designed to compare the performance of Physiologix to human serum as a supplement to support T cell growth, expansion, and viability while increasing transduction efficiency.
eBook:
Purification of antibody therapeutics – DOWNLOAD EBOOK
 Approach for diversified pipeline of multispecific antibodies and antibody fragments
Approach for diversified pipeline of multispecific antibodies and antibody fragments
Antibody therapeutics are the largest class of biotherapeutics, and their development has been ongoing for decades. Over the years, development beyond traditional monoclonal antibodies (mAb) has increased, like multispecific and bispecific antibodies, antibody fragments, and antibody drug conjugates.
Manufacturing platform approaches conveniently used for many traditional mAbs are being adopted or tweaked to fit the respective molecule.
In the eBook Purification of antibody therapeutics, you will learn about considerations and tips for developing purification protocols for antibody variants.
Download eBook
eLearning Opportunity:
DESIGN OF EXPERIMENTS (DOE) FOR CHROMATOGRAPHY – TakE FREE E-LEARNING COURSES
Improve process development efficiency compared to studying one factor at a time (OFAT)
A Design of Experiments (DoE) model can be used to understand how experimental parameters affect outcomes. In  chromatography process development, a DoE approach can greatly improve the efficiency in screening for suitable experimental conditions, in optimizing a process, or in robustness testing.
chromatography process development, a DoE approach can greatly improve the efficiency in screening for suitable experimental conditions, in optimizing a process, or in robustness testing.
What is DoE?
Design of experiments (DoE) is a technique for planning experiments and analyzing the information obtained. The technique allows you to use a minimum number of experiments. You systematically vary several experimental parameters simultaneously to obtain sufficient information to create a mathematical model of the chromatography process.
Take one of the DoE eLearning courses from Cytiva
Explore the basic concepts of DoE, learn about the typical workflow, and get guidance on graph interpretation in the first course. Then see how to perform a DoE screening with UNICORN™ software to optimize conditions for high yield and protein purity.
Headlines:
“Amgen expands pact with Amazon to usher drug manufacturing into the AI era,” FiercePharma
Seeking to identify ways to improve the discovery and production of medicines, Amgen is expanding its partnership with tech giant Amazon. Specifically, Amgen is growing its decade-old collaboration with Amazon Web Services (AWS)—a widely used cloud platform—to create generative AI in a bid to increase the manufacturing throughput of pharmaceuticals. The companies aim to use the tech to boost operations and sustainability at Amgen’s upcoming assembly and final product packaging facility, which is slated to open next year in the greater Columbus, Ohio, area…
“WHO authorizes emergency use of Novavax’s updated COVID shot,” Reuters
Novavax’s (NVAX.O) updated vaccine has been granted emergency-use authorization by the World Health Organization (WHO) for active immunization to prevent COVID-19 in individuals aged 12 and older, the company said on Tuesday. The updated Novavax shot, which was authorized in the U.S. last month, targets a descendant of the XBB lineage of the coronavirus that was globally predominant earlier this year…
“Thermo Fisher teams up with Flagship Pioneering to forge new life science tool startups,” FierceBiotech
Thermo Fisher Scientific has had a longstanding agreement with Flagship Pioneering to help supply the incubator’s roster of young biotechs with life sciences hardware and research services. Now, Thermo Fisher is getting in on the startup game itself. The two have expanded their partnership with plans to work together on forming, developing and commercially scaling what they described as “new platform companies focusing on novel tools and capabilities” aimed at supporting first-in-class therapies, according to their announcement…
“Lilly Puts Potential $660M on the Line for Protein-Protein Interaction Blockers,”BioSpace
Eli Lilly on Tuesday signed a license and collaboration agreement with Tokyo-based biotech PRISM BioLab to discover and develop small molecule inhibitors of protein-protein interactions. Under the terms of the deal, Lilly will make an upfront payment and pledges up to $660 million in preclinical, clinical and commercial development milestones. The Japanese biotech will also be eligible for royalties on future sales of any pharmaceutical product that emerges from the partnership…
“2023’s Fiercest Women in Life Sciences,” FiercePharma
In many ways, 2023 has been a banner year for women. The blockbuster success of the Barbie movie ushered in a tsunami of girl power, women’s sports achieved new viewership highs during the Women’s World Cup and WNBA Finals, and female artists took center stage in both the recently announced Grammy Award nominations and chart-topping world tours led by Taylor Swift and Beyoncé—to name just a few major milestones. That’s not to say that the pervasive inequities that have long plagued every level of power are suddenly gone. According to a recent report from London-based life sciences staffing company Meet Recruitment, which surveyed more than 4,600 life sciences workers around the world, only about a third of C-suite positions are held by women, and more than 40% of respondents said they believe men still have a leg up when it comes to career development opportunities like promotions and raises…
“Valneva says EU regulator accepts its chikungunya vaccine application,” Reuters
French drugmaker Valneva (VLS.PA) said on Monday that the application for its vaccine candidate against mosquito-borne viral disease chikungunya was accepted by the European Medicines Agency (EMA) which would now examine the drug in a fast-track procedure. The EMA “has determined that all essential regulatory elements required for scientific assessment were included in the application,” Valneva said in a statement…