
How to minimize contamination risk and protect your cultures
Company: Corning
Job Title: Scientific Supporting Specialist supporting EMEA regions
Dr. Elisabetta Difilippo is Scientific Supporting Specialist supporting EMEA regions at Corning Life Sciences. She assists in inquiries regarding products and applications of Corning Life Sciences portfolio, and she conducts scientific trainings. Dr. Difilippo graduated in Pharmaceutical Chemistry and Technology, and she received her doctorate in Food Chemistry. She has been with Corning for 4 years.
Dr. Eva Nokes is the Manager of the North American Scientific Support group at Corning Life Sciences. She received her Ph.D. in Molecular and Cellular Biology from Brandeis University. Dr. Nokes supports the Corning Life Sciences portfolio, assisting customers with product selection, application support and troubleshooting. She has been with Corning for 5 years.
Laboratory and cell culture contamination is a serious risk for research labs, process development labs and biomanufacturing. Contamination can compromise cell culture and experiment results, thus wasting valuable resources conducting experiments that ultimately aren’t accurate. It is also of great concern to biomanufacturing as it is one of the leading causes of batch failure.
Unfortunately, contamination is a routine threat. Contamination can be introduced through human interaction, cross-contamination with an already infected culture, poor lab techniques, and contaminated raw materials, just to name a few. In addition, contamination isn’t always easy to detect, which means that contamination can spread to multiple cultures before the problem is identified.
With contamination lurking everywhere, it may feel daunting to think about protecting precious cultures. Not to worry, help is here! For this Ask the Expert Session, we have assembled a team of experts to answer your questions on contamination and minimizing risk. Please see our experts’ bios below.
Question 1
We are in process development on using a closed system to produce adherent cells. What steps would you take to be sure you are starting with a contamination free culture before putting it into the closed system. Also would you test during culture or wait until harvest. At what points in production process do you advise people to test for contamination?
Bioburden control is required/crucial for key applications, as example during pharmaceutical drug manufacturing processes.
Aside from testing raw materials and media sterility at the very beginning of the process, some “in-process testing” can be suggested as below:
- Sterility test every time the culture is passaged.
- Sterility test of inoculum before inoculating any vessels (or large scale bioreactor that is used as final production step).
- Have a back-up cultures for the seed train process (seed train process = generation of an adequate number of cells for the inoculation of a production bioreactor. Ref: Seed train optimization for suspension cell culture. T. Hernández Rodríguez et al. BMC Proc. 2013; 7(Suppl 6): P9.)
Typically, a small sample of the culture can be taken and inoculated within trypticase soy broth tube (or agar).
These tests are performed during 14 days at 37 and 25 degrees Celsius. As time frame of 14 days can be not feasible during production processes, test sterility of the inoculum for a minimum of 24 hours can be evaluated on site.
In addition, besides incubator standard procedures to ensure incubator sterility (please refer to question 1 within this page), suggestions can be shared for open operations in the biological safety cabinets (BSC) as below:
- Agar plate testing of operator gloves directly after sterile gloves are put on and before beginning the process
- Environmental testing in the BSC – open agar plates are placed in the BSC during cell culture processing
Below is an image of a holistic approach to microbial control strategies. For a more extensive overview, we like to invite you to revise “L. Anastasia. Microbial control strategies in bioprocessing falling short of assuring product quality and satisfying regulatory expectations. American Pharmaceutical Review 16(2) · March 2013”
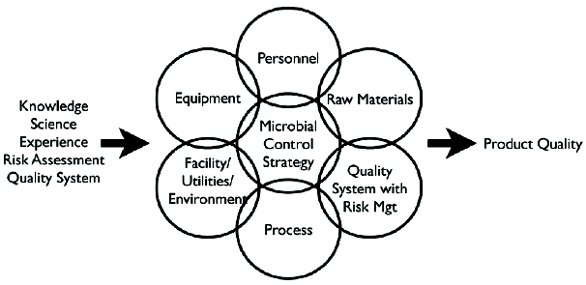
We have many Field Applications Scientists at Corning who have extensive experience with specific bioprocess and closed system approaches. If you have more application specific questions please contact one of the following regional Scientific Support groups to be put in contact with your local Field Applications Specialist. Americas: Scientificsupport@corning.com, Greater Asia (including Greater China, Southeast Asia, Korea, India, Australia and New Zealand): ScientificSupportAP@corning.com, EMEA (including Europe, Middle East and Africa): ScientificSupportEMEA@corning.com, Japan: ScientificSupportJP@corning.com
Question 2
We are culturing Vero cells and we don’t see any obvious signs of contamination, but our media is depleting much more rapidly than it should. Should we test for contamination? If so, can you recommend a broad test to use since we don’t see anything that points us in the direction of a specific contaminant.
There could be two issues. If by “depleting” you mean that the volume of media is going down, then this could be an issue of evaporation. If by “depleting” you are speaking of nutrient depletion, than this could have a few different causes. If this is the case you should check the CO2 and water levels in your incubator to make sure that these are okay. If you exhaust all these possibilities and suspect contamination some quick tests would be:
- Check by PCR and microscopy to make sure that your cells are consistent in their morphology and genetic profile.
- Bacterial: Visual turbidity and pH of the media.
- Endotoxin: Limulus amoebocyte lysate (LAL) assay;
- Mycoplasma: Hoechst stain
Question 3
What are your thoughts on trying to rescue contaminated culture. We recently had a mycoplasma contamination and are debating whether to try and rescue it or just starting over.
This will depend on resources and time. It may be difficult to fully trust the outcome of the experiments after recovery. Also, recovering cells requires time and resources in terms of techniques/media/personnel/equipment to use. At the end of the day the questions are: “Is all the effort worth it? Will I trust the data from this cell culture?” Starting new may take a week or longer, but you will be fully confident on the results and the data can be used for publication. On the other hand, if you have precious primary cells or other limited samples/reagents, then you will probably want to evaluate rescuing.
Question 4
Do you have any advice for ways to prevent contamination beyond good laboratory practices? I think that we do all these things, but we still have the occasional contamination.
Unfortunately even with good laboratory practices the occasional contamination may happen. In order to evaluate action to take regarding occasional contamination, it is important to establish the type of contamination and their origin(s). Sources of contamination and ways of spreading can be very broad, varying from laboratory personnel to unfiltered air to equipment used. All reagents, incubators, and equipment are possible causes that need to be investigated. There can also be instances of air quality in the building causing issues. I invite you to refer to table present in document at: CLS-CGTS-077 https://www.corning.com/catalog/cls/documents/application-notes/CLS-CG-TS-077.pdf. The table gives a first impression on type of contaminations, possible sources, and troubleshooting approaches.
Question 5
We had eliminated the use of antibiotics in our lab, but after a recent contamination, our new lab manager recommended we start using antibiotics for some of our more long term cultures. Do you think this is a good idea?
Antibiotics are extensively used preventive measures to avoid bacterial contamination within in vitro culture. Studies, however, have shown that antibiotics may induce changes in cell gene expression and regulation. For this reason, it is important to understand, test, and evaluate the effect of antibiotics within the specific cell culture outcome. You may also want to evaluate the use of antibiotics if you are doing any compound testing as drugs may interact with antibiotics in unknown ways.
An opportunity for a first testing and evaluation of the effect of antibiotics on the cell gene expression is offered by techniques such as RNA-seq and ChIP-seq. We suggest therefore to evaluate on a case by case basis the use of the antibiotics in short and long term cell cultures. The reference below may offer some guidance on this topic:
Ryu, A.H., Eckalbar, W.L., Kreimer, A. et al. Use antibiotics in cell culture with caution: genome-wide identification of antibiotic-induced changes in gene expression and regulation. Sci Rep 7, 7533 (2017).
Question 6
Do you have any technology that allows you to culture in the incubator and completely eliminate the water tray and eliminate microbe-supporting humidity during your cell culture? What can you do to eliminate transfers for centrifugation steps and for cryopreservation into cryovials? Do you have any cell culture technology that eliminates transfers by spinning directly and cryopreserving directly in the original cell culture container?
These questions are outside of the general cell contamination scope of this forum but we are happy to help you. These technology specific questions would be better answered by a Corning specialist who could find a tailored process based solution for you. Please contact one of the following regional Scientific Support groups and one of our agents can help you or put you in contact with a Field Applications Specialist. Americas: Scientificsupport@corning.com, Greater Asia (including Greater China, Southeast Asia, Korea, India, Australia and New Zealand):ScientificSupportAP@corning.com, EMEA (including Europe, Middle East and Africa): ScientificSupportEMEA@corning.com, Japan: ScientificSupportJP@corning.com
Question 7
We want to test our cultures for sterility but there doesn’t seem to be an easy way to do this. Are you aware of any rapid tests for sterility like you see for endotoxin?
Typical tests of sterility require a long incubation (14 days) period to determine growth of contaminants. There are some more rapid tests available on the market that will give you results in less time, sometimes as short as three days. However, we have not internally tested these methods so unfortunately cannot give you guidance into their validity or performance. You will need to set up a testing procedure that is relevant to your specific needs. Below is a link to a 2015 review article comparing some method types and considerations for each. https://journals.sagepub.com/
Question 8
Our lab conducts research using MSCs and we buy our media from a supplier. Internally we have been sharing information that we have seen at conferences where there can be contamination in raw materials and media. Do you recommend additional testing for media or other raw materials that you bring into the lab and which tests would you recommend.
Suppliers of cell lines and raw material should conduct extensive testing before releasing a product, and provide documentation listing what tests were done, and indicate the level of sterility. One thing to note is that sterility assurance levels (SAL) may vary by manufacturer or product. SAL is the probability of one random contaminant occurring on a surface after sterilization and it is expressed as 10-n. Example: SAL 10-3 means that there is a probability of 1 contaminated product out of 1,000. That being said, if you have extremely sensitive cells and/or assays, you may choose to filter sterilize your media prior to use. No further testing should be required. However, some laboratories may need have more stringent guidelines and testing should be tailored to those more extensive needs. Our suggestion is to always contact the supplier for more information or clarity about the tests that were performed on the product supplied Note: If you have specific concerns about a media or reagent, it is good practice to make the manufacturer aware of the potential issue so they can conduct the appropriate quality investigation.
Question 9
In our group we always use aseptic techniques and fresh media but we still have bacterial contamination, with media turbidity and pH decrease. Where can it come from?
Often cabinets and incubators can be a source of contamination since these are often shared among multiple people/research groups. It is therefore important to have regular maintenance and cleaning of these. Incubators should be cleaned with Lysol and 70% EtOH once a month and shelves should be autoclaved. Clean water tray often (autoclaved, distilled water) and clean spills immediately. Daily cleaning of hood space with 70% EtOH, as well as monthly cleaning with 10% bleach or equivalent products is suggested.
Question 10
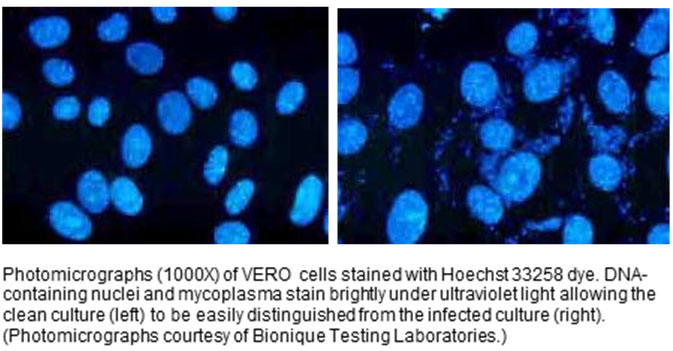
How can I spot mycoplasma contamination?
Mycoplasma contamination is indeed a key factor to evaluate during a cell culture projects, as it can influence cell behavior, morphology and function. Moreover, you may be asked for mycoplasma testing results by journal article reviewers (example: https://www.nature.com/nature-research/editorial-policies/reporting-standards). Fortunately, there are simple fluorochrome DNA staining tests, as well as PCR based mycoplasma tests, that can detect mycoplasma. These can be done simply in your lab, or your samples can be sent out to an outside lab for testing.
Question 11
We have a protocol for freezing cells in our lab, but we have some contamination. Do you have a checklist you can recommend to reduce the possibility of contamination in frozen cell stocks.
BEFORE CRYOPRESERVATION:
- First ensure that the cells are in their best possible condition. Select cultures near the end of log phase growth (approximately 90% confluent) and change their medium 24 hours prior to harvesting. Carefully examine the culture for signs of microbial contamination. Facilitate this by growing cultures in antibiotic-free medium for several passages prior to testing. This allows time for any hidden, resistant contaminants (present in very low numbers) to reach a higher, more easily detected level. Samples of these cultures are then examined microscopically and tested by direct culture for the presence of bacteria, yeasts, fungi, and mycoplasmas.
- Mycoplasmas present a special problem since they can be found in cultures at very high concentrations (up to 108 organisms per milliliter of medium) without any visible effects or turbidity. As a result, as many as 20% of all animal cell cultures are contaminated by these ubiquitous but unseen organisms. Although special efforts are required to detect mycoplasmas, the serious consequences of their presence makes testing frozen culture stocks absolutely essential
- Use equipment that will help to stabilize temperature and decrease temperature fluctuation. Corning’s sample cooling and heating solutions enable consistent, reproducible, standardized temperature control with or without ice, electricity, or batteries. These solutions address the pitfalls of temperature regulation and reduce contamination risk, keeping your samples cool and stable while you work. https://www.corning.com/catalog/cls/documents/brochures/CLS-BC-CC-082.pdf
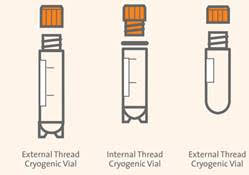
- Store cells in specifically designed cryovials. Several cap styles are available, some with an internally-threaded stopper, and others with externally-threaded designs which help minimize contamination (figure below).
DURING CRYOPRESERVATION:
- Liquid nitrogen (N2(l)) freezers permit storage either in the vapor phase above the liquid at temperature between -140°C and -180°C, or submerged in the liquid at a temperature below -196°C. Using vapor phase storage greatly reduces the possibility of leaky vials or ampules exploding during removal, therefore it can be suggested to prevent spread of contamination.
- More information about cryopreservation, are present at: https://www.corning.com/worldwide/en/products/life-sciences/resources/webforms/we-get-you-guide-for-cryogenically-storing-animal-cell-cultures.html
- Please also see checklist for freezing cell cultures: https://www.corning.com/
catalog/cls/documents/posters/ CLS-CC-111.pdf.
Question 12
We haven’t had this happen in our lab, but I am curious what do you do if you have contamination to make sure that you’ve gotten rid of the source of the contamination?
If you experience contamination and thoroughly clean your incubators, water baths, hoods and other work surfaces, and also change any media or reagents that could potentially have been contaminated, you will likely have gotten rid of the source. You may never know exactly what the source was, and it might not be necessary to, but for a few weeks after you should keep a close eye on your cultures and make sure all lab personnel know and follow safe handling processes. It is possible someone noticed that a bottle of media fell over in the water bath and “thought it would be fine” or were in a hurry and neglected to put on their gloves or spray down their work surface, or answered their phone while changing media. After a major contamination event that person will suspect they may have been the cause and there will not be a repeat accident. Most researchers will occasionally take shortcuts in the lab, and find out the hard way that some of these can lead to major problems. If you do notice a second round of contamination close to cleaning up after the first you may need to retrain all of your lab personnel, as well as re-check all media, reagents, and frozen cultures to determine with certainty what the cause was. Communication is essential for working with multiple users in shared cell culture spaces.
Question 13
I haven’t seen a question yet on cell line contamination, i.e. the cells don’t have the correct lineage. What are your thoughts on how to deal with this problem?
Cross-contamination of cultures is the unintended mix up of different cell lines leading to a mixed population . It can have various causes as example: cross-use or sharing of media from a different cell line, and share Equipment (Biosafety cabinets).
Approaches :
- Quarantine all cultures coming into the laboratory until they have been tested/checked first. A first step when starting a culture is cell authentication to determine identity and species of cells
- Periodically check your cells for morphology or genetic changes. Cell authentication to determine identity and species of cells is extremely important
- Work with only one cell line at a time whenever possible
- Thoroughly clean before and after introducing a new cell line into the laminar flow hood
- Daily cleaning of hood space with 70% alcohol, as well as monthly cleaning with 10% bleach or equivalent products
- Storing cell line stocks in vapor phase of LN2 , not in liquid phase
Ref: CLS-CG-TS-077 REV1 https://www.corning.com/catalog/cls/documents/application-notes/CLS-CG-TS-077.pdf
Please also see - A guide to contamination, which can be a helpful resource as well: https://www.corning.com/